Gluconato Cúprico
-
Nombre del producto:
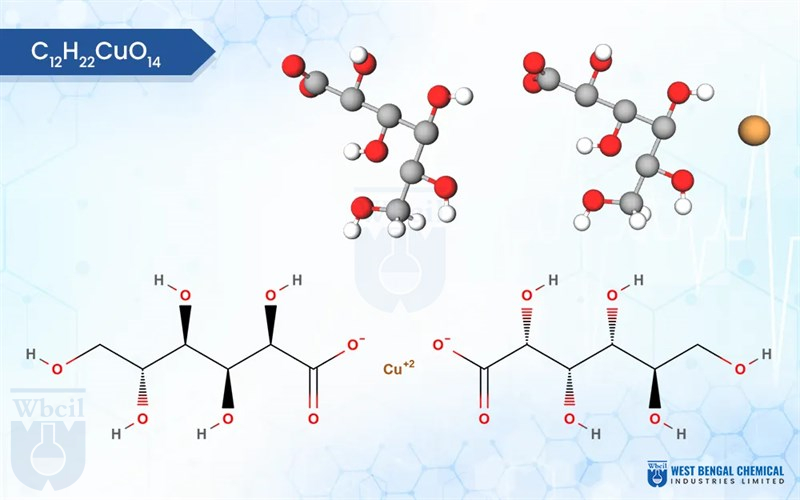
Gluconato Cúprico
-
Fórmula molecular:
C12H22CuO14
-
Peso molecular:
453.84 g/mol
-
CAS No.:
13005-35-1
-
Código HSN:
29181690
-
Código CID:
10692
-
Vida útil:
3 años - 20°C polvo
-
Código ATC (WHO)
V03AB20
-
DrugBank ID
DB11246
-
ChemSpider ID
10242
-
UNII No.
RV823G6G67
- USP
- IUPAC Names
- Sinónimos
USP of Gluconato Cúprico
- El gluconato de cobre es conocido por su biodisponibilidad relativamente alta.
- El gluconato de cobre está disponible en varias formas, como comprimidos, cápsulas y suplementos líquidos.
IUPAC Names of Gluconato Cúprico
- copper;(2R,3S,4R,5R)-2,3,4,5,6-pentahydroxyhexanoate
- Copper(II) gluconate
Sinónimos de Gluconato Cúprico
- Copper gluconate
- Copper(II) gluconate
- Cas: 527-09-3
- Copper(II)gluconate
- Cupric Gluconate
- Copper D-gluconate (1:2)
- Copper(2+) D-gluconate, (1:2)
- UNII-RV823G6G67
- Bis(D-gluconato-O1,O2)copper
- Gluconic acid, copper(2+) salt
- Bis(D-gluconato)copper
- RV823G6G67
- Copper, bis(D-gluconato-O1,O2)-
- Copper(II) (2R,3S,4R,5R)-2,3,4,5,6-pentahydroxyhexanoate
- Copper (as Gluconate)
- Gluconic acid, copper(2+) salt (2:1), D-
- Copper, bis (D-gluconato-O(sup 1),O(sup 2))-
- copper;(2R,3S,4R,5R)-2,3,4,5,6-pentahydroxyhexanoate
- 13005-35-1
- Copper di-D-gluconate
- Gluconates
- Gluconic Acid Copper(II) Salt
- Gluconate, Copper
- Cupric gluconate monohydrate
- Copper, bis(D-gluconato)-
- Chelates of copper gluconate
- Copper,bis(D-gluconato-kO1,kO2)-
- Copper, bis(D-gluconato-kappaO1,kappaO2)-
- Gluconic acid, copper(2+) salt (2:1) D-
Si está interesado en Gluconato Cúprico, entonces
Descripción de Gluconato Cúprico
El gluconato de cobre es una sal cúprica del ácido glucónico, y se utiliza habitualmente como suplemento dietético para aportar el mineral esencial cobre al cuerpo humano. El gluconato de cobre contiene alrededor de un 14-15% de cobre elemental.
Si está interesado en Gluconato Cúprico, entonces
Aplicación de Gluconato Cúprico
Como suplemento dietético, es ampliamente popular como seguro para el consumo a un nivel máximo del 0,005 por ciento.
Productos Relacionados
Si está interesado en Gluconato Cúprico, entonces