Bioavailability of Liposomal Coenzyme Q10: Why Standard CoQ10 Fails
The bioavailability of liposomal Coenzyme Q10 has become a strategic consideration for nutraceutical formulators and brand owners seeking clinically credible performance. Many finished products underperform not because of inadequate dosage but because of the inherent absorption limitations of standard crystalline CoQ10.
Its lipophilic structure and poor aqueous dispersion create measurable inefficiencies across the gastrointestinal and hepatic absorption pathway. As regulatory scrutiny and consumer awareness increase, delivery technology now differentiates premium formulations from commodity offerings.
In this blog, we analyse the absorption barriers, clinical pharmacokinetic data, and sourcing considerations that define next-generation CoQ10 supplementation.
Key Takeaways:
- Standard CoQ10 demonstrates low systemic uptake due to its low solubility and limitations in the digestive pathway.
- Liposomal delivery enhances plasma exposure without proportional dose escalation.
- Precision in manufacturing and vesicle stability determine measurable clinical performance outcomes.
Quick Answer: Liposomal delivery improves CoQ10 absorption by protecting the molecule and increasing measurable systemic exposure without higher dosing.
Why CoQ10 Is Naturally Hard to Absorb
The bioavailability of Lipoedge liposomal coenzyme Q10 becomes relevant only after understanding why standard CoQ10 struggles to absorb in the first place.
- Large, highly lipophilic molecule: CoQ10 is a fat-soluble benzoquinone with a long isoprenoid tail, making it extremely hydrophobic and poorly miscible in water-based gastrointestinal fluids. Its size and structure limit passive diffusion across intestinal membranes.
- Poor Solubility of CoQ10: In its native state, CoQ10 exhibits negligible aqueous solubility, requiring incorporation into mixed micelles with bile salts before absorption can begin. Without adequate dispersion, intestinal uptake remains minimal.
- Ineffective Crystalline Form of CoQ10: Commercial CoQ10 commonly exists in crystalline polymorphic forms (Form I/II). These tightly packed crystal lattices must fully dissociate into single molecules before absorption; incomplete crystal dispersion significantly limits uptake efficiency.
- Dispersion barrier reduces absorption: Mantle & Dybring report that failure to disperse CoQ10 crystals in the gut adequately can reduce bioavailability by up to 75%, highlighting crystal dissolution as a critical bottleneck [1].
- Hostile gastrointestinal environment: The aqueous intestinal lumen, variable bile secretion, and reliance on lipid digestion pathways collectively contribute to the low bioavailability of CoQ10, especially when delivered without formulation technology.
Also read: All You Need to Know About WBCIL’s Liposomal CoQ10.
3 Stages Where Standard CoQ10 Loses Effectiveness
To fully appreciate the bioavailability of liposomal coenzyme Q10, it is important to understand where standard CoQ10 fails across the natural absorption pipeline.
Here are three stages where conventional CoQ10 loses its efficacy:
Stage 1: Gastric & Intestinal Dispersion Failure
After oral intake, CoQ10 must first dissolve in the gastrointestinal tract before it can be absorbed, which makes bypassing the stomach an important consideration in advanced delivery systems. Because of CoQ10’s low aqueous solubility due to its hydrophobic crystalline structure, dispersion in the aqueous gut environment remains highly inefficient. Without formulation support, much of the ingested CoQ10 stays undissolved and passes through the digestive tract without meaningful absorption.
Stage 2: Hepatic First-Pass Limitation
Even after successful intestinal uptake, absorbed CoQ10 enters chylomicrons and travels through lymphatics before reaching systemic circulation. A portion is metabolised during hepatic processing, reducing circulating plasma concentrations. Industry analysts note that significant losses occur during this metabolic filtering phase, contributing to variability in blood-level responses among individuals.
Stage 3: Lymphatic Absorption Bottleneck
As a lipophilic compound, CoQ10 depends on dietary fat and bile salts for micellarisation. In fasted conditions, micelle formation is limited, resulting in poor systemic uptake. CoQ10 reduces to its ubiquinol form during or after the absorption processes, and 95% of it circulates within the body [2].

Next, let’s explore the mechanism of liposomal absorption with reference to its bioavailability.
Mechanism of Liposomal Absorption Explained
The bioavailability of liposomal coenzyme Q10 improves because liposomes change how the molecule interacts with biological membranes rather than simply protecting it during digestion.
Here are some of the key considerations to understand the liposomal delivery and absorption:
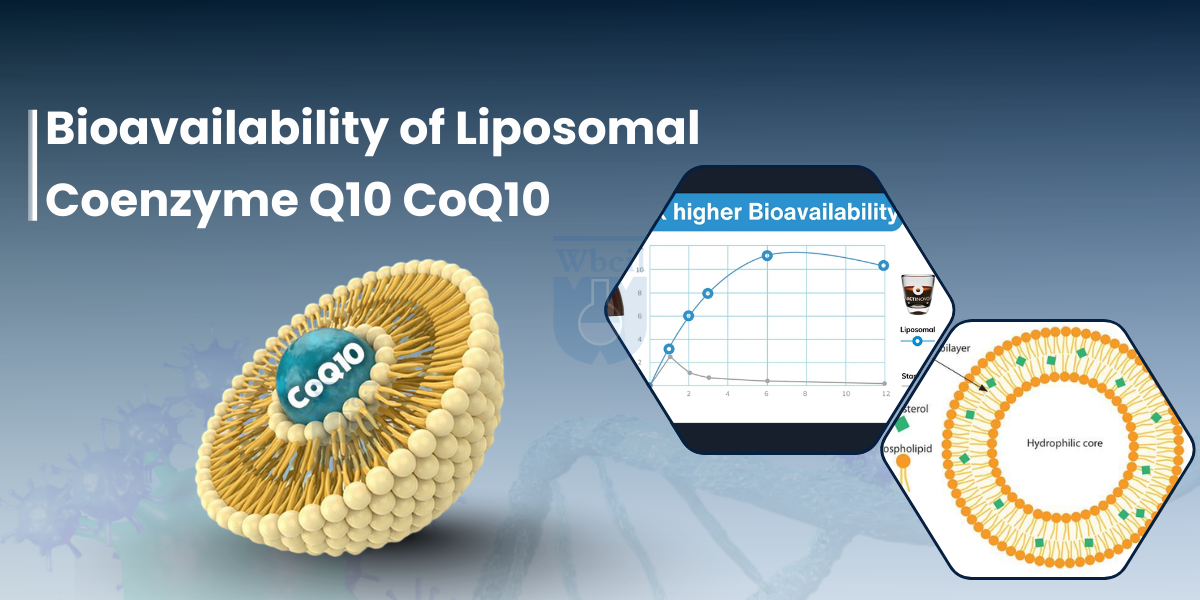
- Membrane-Mimicking Architecture: Liposomes are nano-sized (50–200 nm) phospholipid vesicles that structurally resemble human cell membranes. The similarity allows them to integrate more naturally with intestinal epithelial cells compared to free crystalline CoQ10.
- Enhanced Dispersion at the Molecular Level: Instead of relying on bile-mediated micelle formation, liposomal encapsulation keeps CoQ10 in a pre-dispersed state. It directly addresses the poor solubility of CoQ10 and prevents recrystallisation in the gut lumen.
- Improved Transcellular Transport: Liposomes can undergo endocytosis or membrane fusion, enabling transport across intestinal barriers without complete dependence on dietary fat intake. It reduces variability linked to fed vs fasted absorption.
- Direct Cellular Delivery: Once in circulation, liposomes may fuse with target cell membranes, allowing intracellular release of CoQ10 in energy-demanding tissues such as myocardium and skeletal muscle.
- Faster and Higher Peak Concentration: Pharmacokinetic trials report significantly higher plasma exposure with more than 31.3% for liposomal CoQ10, supporting its role as the best delivery system for CoQ10 supplements in advanced liposomal nutraceuticals [3].
Now, let’s explore some of the clinical evidence to ascertain the bioavailability of liposomal CoQ10 in industry settings.
Clinical Evidence on Liposomal CoQ10 Bioavailability
Accurate data and clinical trials now confirm that the bioavailability of liposomal coenzyme Q10 is measurably superior to that of standard crystalline formulations when engineered correctly.
Here are some of the key pharmacokinetic outcomes from controlled clinical research:
| Parameter | Liposomal CoQ10 Result | Clinical Interpretation |
| Cmax (Maximum Blood Concentration) | 31.3% higher vs standard CoQ10 (p < 0.001 ) [4] | Demonstrates Enhanced Absorption of CoQ10 and higher efficacy ceiling |
| AUC₀–24 (24-hour Exposure) | 22.6% greater systemic exposure (p < 0.001) [5] | Indicates sustained plasma availability over time |
| Tmax (Time to Peak) | Achieved without a fatty meal requirement | Faster and Higher Peak Concentration with reduced fed-state dependency |
These pharmacokinetic improvements were demonstrated in a double-blind, randomised controlled trial, strengthening the clinical credibility of liposomal delivery systems.
Importantly, broader pharmacokinetic analyses emphasise that formulation quality remains critical. Reviews indicate that poorly engineered liposomal APIs may fail to enhance uptake, reinforcing that vesicle stability, phospholipid composition, and particle size control determine real-world performance.
Liposomal vs Standard CoQ10: What Works Better?
When evaluating liposomal vs standard CoQ10, formulators must move beyond label claims and examine how delivery technology influences performance, cost structure, and product positioning.
Here is a comparative representation to understand the difference between the two forms:
| Parameter | Standard CoQ10 | Liposomal CoQ10 |
| Absorption Rate | Typically, 10–15% systemic uptake under conventional conditions | Significantly higher plasma exposure demonstrated in controlled trials |
| Meal Dependency | Requires a fatty meal for optimal micelle formation | Not strictly dependent on dietary fat |
| Stability Profile | Crystalline structure prone to aggregation; sensitive to oxidation | Phospholipid encapsulation enhances dispersion and protects actives |
| Dose Efficiency | Higher dose required to achieve target plasma levels | A 30-40% lower dose may achieve comparable systemic exposure |
| Formulation Versatility | Limited to oil-based soft gels and capsules | Dispersible in aqueous formats (sachets, beverages, gummies) |
Also read: From Lab to Label: The API Journey from Manufacturing to Market.
Why WBCIL’s Liposomal CoQ10 Leads in B2B Sourcing
Transitioning from clinical promise to commercial success requires a partner with proven manufacturing and regulatory capability. WBCIL’s Liposomal CoQ10 is produced in WHO-GMP and ISO-certified facilities, backed by more than six decades of API and fine chemical expertise. Its liposomal platform is designed to address the low absorption of CoQ10 associated with conventional crystalline formats while supporting scalable B2B supply.
For brand owners and formulators, WBCIL offers flexible integration into capsules, sachets, liquid suspensions, and gummies. Technical specifications and documentation are available through the company’s liposomal products to support regulatory and formulation decisions. Nutraceutical companies seeking reliable, high-performance CoQ10 ingredients can request data sheets, stability information, and sampling for evaluation.
Final Thoughts
For B2B formulators, delivery technology must be evaluated alongside raw material quality and regulatory documentation. Pharmacokinetic metrics such as Cmax, AUC, and stability under storage conditions should guide formulation decisions.
Published data support improved systemic exposure when the bioavailability of liposomal Coenzyme Q10 is engineered correctly. Procurement teams should assess encapsulation integrity, phospholipid quality, and batch reproducibility before supplier on-boarding. Nutraceutical companies can collaborate with WBCIL to align formulation science with scalable, compliant production.
- https://pmc.ncbi.nlm.nih.gov/articles/PMC7278738/
- https://www.ncbi.nlm.nih.gov/books/NBK531491/
- https://pmc.ncbi.nlm.nih.gov/articles/PMC12486408/
- https://www.frontiersin.org/journals/nutrition/articles/10.3389/fnut.2025.1605033/full
- https://pubmed.ncbi.nlm.nih.gov/41041129/
Clinical comparisons show that delivery format significantly influences absorption. Liposomal CoQ10 formulations have demonstrated improved plasma exposure compared to standard crystalline CoQ10, particularly when properly engineered for stability and particle size control. Bioavailability ultimately depends on formulation quality rather than dosage strength alone.
Liposomal CoQ10 can offer improved systemic exposure by enhancing dispersion and reducing digestive losses. Standard crystalline CoQ10 often exhibits low absorption due to poor solubility and reliance on fat-dependent uptake pathways. However, performance varies based on manufacturing precision and encapsulation integrity.
Ubiquinol is the reduced form of CoQ10 and may be better absorbed in certain populations. Liposomal CoQ10 focuses on delivery enhancement rather than molecular conversion, improving uptake through membrane-compatible transport. Comparative performance depends on formulation design, clinical objectives, and target demographic.
Export-ready liposomal CoQ10 requires GMP-compliant manufacturing, validated stability data, and regulatory documentation. WBCIL (West Bengal Chemical Industries Limited), with its expertise in liposomal platforms, typically supports international private-label and bulk supply. Buyers, however, should verify the consistency of encapsulation, the sourcing of phospholipids, and batch traceability before procurement.
Standard CoQ10 often depends on dietary fat for micelle formation and absorption efficiency. Liposomal delivery systems are designed to reduce meal dependency by improving dispersion and membrane interaction. This can help provide more consistent plasma levels across varying dietary conditions.











