Potassium Glycinate in Nutraceutical: Metallic Taste Control
Metallic and bitter notes are a known sensory issue in the liquid system. Recent studies suggest that the threshold for metallic perception can be as low as 0.06% for simple potassium salts in solution, which can affect consumer acceptability in oral products [1]. Even advanced flavour systems cannot fully mask the sensory impact of free ionic potassium in solution.
In potassium glycinate in nutraceuticals, chelation offers a structured approach to improve palatability and formulation control. Taste stability, regulatory compliance, and shelf performance directly influence procurement approval and repeat purchase behaviour.
In this blog, the scientific, regulatory, and sourcing aspects of chelated potassium for syrup systems are examined from a formulation perspective.
Request Technical Information for Potassium Glycinate (Specs, Uses & Quality Standards)
Key Takeaways:
- Free ionic potassium contributes to metallic perception and reduced adherence in liquid nutraceutical syrups.
- Chelation reduces ionic reactivity, supporting improved taste stability and precipitation control.
- WHO-GMP compliant sourcing strengthens regulatory readiness and long-term commercial viability.
Quick Answer: Chelated potassium improves taste stability, supports shelf performance, and aligns with regulatory requirements for compliant syrup formulations.

Why Liquid Nutraceuticals Develop Metallic Off-Notes
In potassium glycinate in nutraceuticals, taste control determines product success because free potassium ions from inorganic salts trigger metallic and bitter perception in liquid dosage forms.
- Inorganic salts such as potassium chloride dissociate fully in water, releasing free K⁺ ions that activate bitter taste receptors. A study reported that potassium chloride dry syrup was used at 20% w/w concentration, which required taste masking to improve palatability [2].
- Sensory research shows that 100% replacement of sodium chloride with potassium chloride markedly increases metallic perception in panel testing.
- Liquid syrups expose dissolved ions directly to oral receptors, while capsules delay contact until gastric transit, thereby reducing the immediate taste impact.
- Repeated dosing in paediatric and geriatric populations can cause flavour fatigue, reducing adherence to compliance-dependent electrolyte products.
- These challenges drive demand for chelated potassium for syrups. A potassium glycinate chelate reduces free-ion interactions, and potassium glycinate in a syrup formulation supports improved palatability without heavy masking.
Next, let’s understand how chelation helps improve taste with reference to potassium glycinate in nutraceuticals and supplements.
Potassium Glycinate in Nutraceuticals: How Chelation Improves Taste
Chelation changes how potassium behaves chemically in solution, and that shift directly improves palatability and formulation stability in your syrup system.
Here are some key considerations for understanding how chelation improves the efficacy of nutraceuticals.
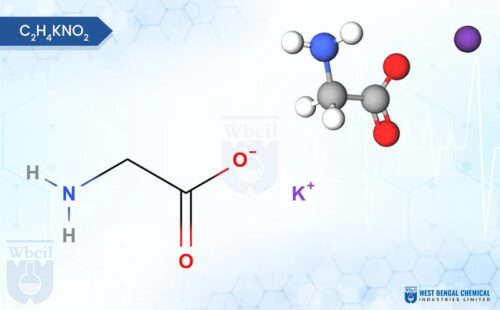
- A potassium glycine chelate forms when one potassium ion associates with glycine molecules, which reduces free ionic interaction in solution. Chelated minerals remain more stable in aqueous media.
- Chelation reduces direct stimulation of bitter taste receptors because potassium does not exist as freely dissociated K⁺ in the same manner as simple salts. This lowers metallic perception in liquid products.
- Potassium glycinate has a low molecular weight well below 500 Daltons, which supports transport through amino acid pathways rather than passive ionic diffusion [3].
- In comparison, potassium chloride dissociates fully and contributes stronger bitterness, while potassium gluconate improves tolerance but still releases free ions.
- In potassium glycinate in syrup formulation, reduced ionic reactivity improves compatibility with sweeteners, stabilisers, and acidic excipients.
- For B2B formulators, chelation supports taste neutrality, predictable bioavailability, and improved gastrointestinal tolerance without reliance on heavy flavour masking systems.
Also read: The Role of Mineral Chelates in Preventing Nutrient Deficiencies.
Top 5 Formulation Benefits of Potassium Glycinate in Syrups
Potassium glycinate offers five distinct formulation benefits that matter most when you work with aqueous syrup systems at scale.
Here are five benefits for potassium glycinate in nutraceuticals:
1. Solubility and Molecular Stability in Aqueous Media
Chelated potassium remains associated with glycine in solution, which moderates free ionic dissociation. This reduced ionic activity limits unwanted interaction with acidic components in syrup matrices. PubChem lists potassium glycinate with a molecular weight of 113.13 g/mol, which supports rapid dissolution in aqueous systems [4].
2. pH Compatibility and Precipitation Control
Liquid nutraceutical syrups typically operate between pH 3.0 and 5.5. In this acidic range, simple salts may show stronger ionic interaction and taste impact. Chelation supports shelf-life and precipitation control by reducing uncontrolled ion exchange reactions.
3. Compatibility with Common Syrup Excipients
Sorbitol, sucralose, xanthan gum, and citric acid form common syrup bases. Excess ionic reactivity may alter clarity or mouthfeel in such systems. Glycine chelation reduces reactivity and supports flavour fatigue reduction in repeated-dose formulations.
4. Sodium-Free Electrolyte Positioning
Electrolyte syrups often target sodium-sensitive groups. Potassium glycinate provides sodium-free electrolyte support without adding Na⁺ load. The positioning supports formulations designed for electrolyte balance and mild mineral deficiency correction.
5. Stability Framing Under ICH Zone IVb
ICH climatic Zone IVb requires long-term stability testing at 40°C ±2°C and 75% RH ±5% RH for tropical markets. These parameters are defined in WHO Technical Report Series No. 953, Annexe 2 [5]. Glycine chelation benefits matter under such stress conditions because controlled ionic behaviour supports consistent assay and physical stability over labelled shelf life.
Now that we have a better understanding of the benefits of potassium glycinate, let’s explore how it offers greater efficacy and commercial value than conventional potassium salts.

Potassium Glycinate vs Other Potassium Salts: A B2B Comparison
Your choice of potassium salt affects taste neutrality, absorption behaviour, regulatory acceptance, and long-term commercial performance in liquid dosage forms.
Here is a tabular representation to understand the difference between inorganic salts and potassium glycinate to understand the efficacy of potassium glycinate in nutraceutical:
| Parameter | Inorganic Salts (KCl / Citrate / Gluconate) | Potassium Glycinate |
| Taste Profile | Fully dissociate and release free K⁺, which produces bitter and metallic perception in liquids | Chelated structure reduces free ionic interaction, which supports a cleaner sensory profile |
| Ionic Behaviour | High ionic reactivity in acidic syrup systems | Reduced ionic reactivity due to amino acid chelation |
| Bioavailability | Good absorption, but dependent on ionic stability | Amino acid chelated minerals may demonstrate higher bioavailability in controlled comparisons. |
| GI Tolerance | Higher concentration may cause gastric discomfort | Chelated form supports improved gastrointestinal tolerance |
| Sodium Status | Sodium-free | Sodium-free |
| Regulatory & Procurement Fit | Accepted under FSSAI and CDSCO when compliant | Often preferred when sourcing WHO-GMP APIs for nutraceuticals to support audit and dossier requirements |
For procurement teams, elemental content alone does not determine suitability. Sensory acceptance influences repeat usage in paediatric and geriatric segments. Stability under acidic pH affects clarity and dose reliability. Regulatory documentation and GMP status determine supplier credibility in competitive nutraceutical markets.
Also read: From Lab to Label: The API Journey from Manufacturing to Market.
WHO-GMP Compliance for Potassium Glycinate APIs
Regulatory discipline at the API level determines whether your syrup passes audit, holds stability, and protects your brand across regulated markets.
Here are some of the core considerations for WHO-GMP compliance for potassium glycinate APIs:
- WHO-GMP Compliance: Liquid nutraceutical APIs must meet WHO-GMP and current GMP standards to ensure batch consistency and traceability. FSSAI-regulated products require compliant raw materials to avoid regulatory objections. A qualified potassium glycinate API manufacturer should operate under audited quality systems with validated procedures.
- Dossier Requirements: You should verify a complete CoA with assay range, heavy metal limits, and microbial profile. Stability data relevant to tropical climates strengthens regulatory submissions. DMF support and validated analytical methods reduce approval risk.
- Shelf-Life Impact: API quality affects precipitation behaviour, clarity, and dose uniformity in syrup systems. Variability in impurity profile or moisture content may reduce long-term stability. Controlled manufacturing supports predictable 24-month pack performance.
- Application Fit: API characteristics must suit liquid systems, not only tablets or capsules. Control over particle attributes and moisture sensitivity improves excipient compatibility. Strong application and technology alignment lowers reformulation risk.
- Sourcing Risks: Missing validation records or inconsistent documentation increase procurement exposure. Non-GMP sourcing may delay launch timelines and weaken regulatory confidence in nutraceutical markets.
Strategic Considerations for Syrup Manufacturers
When developing liquid nutraceuticals, ingredient selection must support taste stability, regulatory compliance, and consistent shelf performance. In potassium glycinate in nutraceuticals, chelation offers measurable advantages in ionic control, precipitation resistance, and sensory acceptance, which directly influence repeat purchase and brand credibility.
You should partner with suppliers who provide full documentation, alignment with tropical market stability, and transparent quality systems consistent with pharma-grade chelated minerals for syrups. If you seek consistent technical support, validated dossiers, and WHO-GMP-compliant sourcing, you may review WBCIL’s products as part of your supplier evaluation process.
- https://www.frontiersin.org/journals/nutrition/articles/10.3389/fnut.2024.1436113/full
- https://link.springer.com/article/10.1186/s41120-019-0030-z
- https://www.nutritionaloutlook.com/view/understanding-chelated-minerals
- https://pubchem.ncbi.nlm.nih.gov/compound/Potassium-glycinate
- https://academy.gmp-compliance.org/guidemgr/files/WHO_TRS_953_Annex2.pdf
Metallic perception usually arises from free ionic potassium in solution. Taste masking strategies include sweetness modulation, acid balance, and flavour layering, but these may not fully neutralise ionic bitterness. Many formulators instead shift to chelated forms to reduce free ion interaction at the source rather than relying solely on masking systems.
Potassium chloride dissociates fully in water and often produces a strong, bitter, and metallic profile. In contrast, potassium glycinate forms a chelated structure that reduces direct ionic stimulation of taste receptors. This structural difference explains why chelated forms typically demonstrate improved sensory neutrality in syrup systems.
Flavour systems for chelated minerals in syrups often rely on balanced sweetness, mild acidity, and compatible aroma profiles. Sucralose, sorbitol, and fruit-acid blends are commonly selected for electrolyte and paediatric formats. Clean-label taste masking for potassium salts may combine natural sweeteners with controlled pH adjustment to enhance acceptance.
Reducing bitterness in electrolyte RTD shots requires control over ionic strength, sweetness profile, and acid buffering. Chelated minerals reduce free ion exposure, which lowers metallic aftertaste risk. Optimised pH and sweetness strategies for mineral syrups improve perception without excessive flavour load.
Potassium glycinate stability in aqueous solutions depends on controlled pH and excipient compatibility. Chelated structures show improved compatibility with sweeteners and organic acids compared with simple salts. Potassium glycinate compatibility with sweeteners and acids supports clearer syrups and reduced precipitation risk in long-term storage.