Liposomal Glutathione: Immune Restoration in Type 2 Diabetes
Standard care focuses on blood sugar but leaves the immune system at risk. Diabetic patients face high infection rates and slow recovery because their internal antioxidant shield fails. Research shows this deficit results from high glucose levels that break the body’s natural defence cycle.
Liposomal delivery provides a proven path to restore these levels where standard oral options fail. This breakthrough offers a vital tool for brands that develop new health products for this condition.
In this blog, we present clinical data demonstrating why advanced liposomal technology supports the long-term wellness of diabetes patients and why reduced glutathione for Type 2 Diabetes needs attention.
Key Takeaways:
- GSH depletion in Type 2 Diabetes is driven by impaired synthesis, not just increased oxidative consumption.
- Liposomal delivery achieves intracellular GSH repletion that standard oral glutathione formulations cannot replicate.
- API quality directly determines whether a liposomal GSH product reproduces clinically validated immune outcomes.
Quick Answer: Type 2 Diabetes drains glutathione through impaired synthesis and oxidative consumption. Liposomal delivery restores NK cells, Th1 balance, and clinical infection defence.

Why Type 2 Diabetes Depletes Glutathione
Glutathione is the body’s most critical intracellular antioxidant, maintaining immune integrity and cellular redox balance across all tissue types. According to Sekhar et al., diabetic patients exhibit erythrocyte glutathione concentrations up to 73.8% lower than those of healthy adults [1].
Here are some of the critical factors that reduce natural glutathione levels in type 2 diabetes:
- The Polyol Pathway Trap: Hyperglycaemia forces excess glucose through the polyol pathway, consuming NADPH that glutathione reductase needs for recycling. It directly starves the antioxidant recycling system, leading to progressively declining active GSH levels across immune cells. Moreover, the higher the blood sugar, the less the body restores its own antioxidant reserves.
● Impaired Biosynthesis: Chronic hyperglycaemia suppresses GCLC and GSS, the two enzymes that synthesise new glutathione within cells. A PubMed study states that diabetic patients show approximately 51.5% of reduction in glutathione synthesis compared to healthy individuals. Lutchmansingh et al.’s findings indicate that such deficiencies are due to decreased synthesis and higher non-glycaemic utilisation in patients with microvascular complications [2].
● Cysteine Substrate Shortage: Oxidative stress prematurely converts cysteine, the rate-limiting GSH precursor, into inactive forms. It creates a permanent upstream bottleneck that compounds existing enzyme-level impairments in glutathione production.
● Mitochondrial Vulnerability: Mitochondria cannot synthesise glutathione and depend entirely on membrane transporters for supply. Hyperglycaemia progressively impairs these transporters, accelerating beta-cell apoptosis and microvascular complications.
● AGE-Mediated Depletion: Persistent hyperglycaemia generates reactive dicarbonyl compounds that bind directly to glutathione molecules. It drains reserves through a pathway completely independent of oxidative stress.
A glutathione deficit does not remain biochemical for long; it rapidly translates into measurable, compounding failures across the entire immune system.
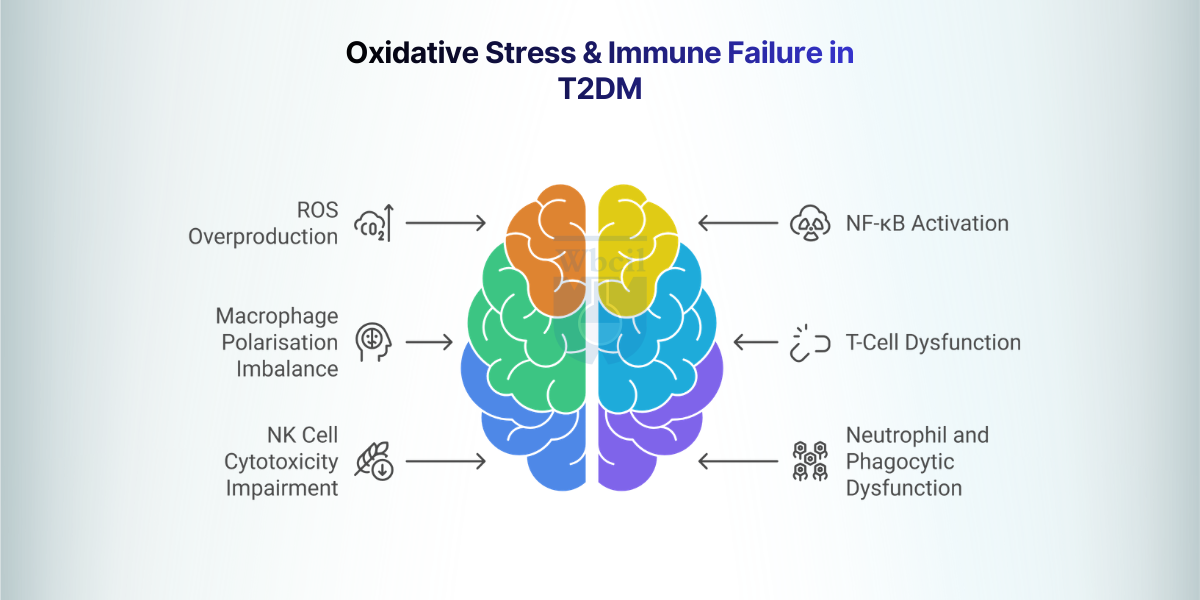
Oxidative Stress and Immune Failure in Type 2 Diabetes
The role of glutathione in Type 2 Diabetes extends beyond antioxidant defence, as it directly governs immune cell function that hyperglycaemia systematically destroys. Chronic oxidative stress in T2DM impairs all major immune cell classes, creating a compounding vulnerability to infection, inflammation, and long-term complications.
Here are six oxidative stress factors and the immune failures for type 2 diabetes:
1. ROS Overproduction: The Initiating Driver
Hyperglycaemia activates multiple simultaneous ROS sources, including mitochondrial uncoupling, NADPH oxidase isoforms NOX2 and NOX4, and xanthine oxidase upregulation. Despite normal β-cell replication, apoptosis is reduced, which ultimately decreases the number of cells by 50% [3]. It creates a chronic pro-oxidant environment that overwhelms the already-depleted GSH-based antioxidant network in diabetic patients.
2. NF-κB Activation and Chronic Inflammation
GSH normally maintains NF-κB in its inactive state by preserving the redox status of IκB kinase within immune cells. With GSH depleted, IKK becomes constitutively active, driving continuous transcription of TNF-α, IL-6, and IL-1β. Such sustained inflammatory signalling simultaneously deteriorates insulin resistance and impairs immune effector function.
3. Macrophage Polarisation Imbalance
Oxidative stress and elevated free fatty acids in T2DM drive macrophage polarisation strongly toward the pro-inflammatory M1 phenotype. M1-dominant macrophages overproduce TNF-α and IL-6, directly blocking insulin receptor signalling in metabolically active tissues [4]. It creates a self-amplifying cycle connecting immune dysfunction directly to worsening insulin resistance.
4. T-Cell Dysfunction and Th1/Th2 Imbalance
GSH maintains the thiol status of key TCR signalling kinases, including Lck and ZAP-70, essential for T-cell activation. With GSH depleted, TCR signal transduction is blunted, reducing T-cell proliferation and protective cytokine output. T2DM patients show significantly reduced IFN-γ:IL-4 ratios, confirming a measurable shift away from protective Th1 immunity.
5. NK Cell Cytotoxicity Impairment
Natural Killer cells provide first-line immune surveillance without prior antigen sensitisation, making their function critical in immune defence against diabetes. Oxidative modification of NK surface receptors NKG2D and NKp46 reduces their ability to recognise and bind infected target cells. T2DM patients exhibit a 3-fold higher susceptibility to tuberculosis, directly reflecting measurable functional failure of NK cells and T cells.
6. Neutrophil and Phagocytic Dysfunction
Neutrophil chemotaxis, phagocytic capacity, and oxidative burst precision are all significantly impaired in Type 2 Diabetes. GSH depletion dysregulates the spatial control of neutrophil ROS production, converting a targeted defence into indiscriminate inflammatory tissue damage. Exaggerated neutrophil extracellular trap formation further amplifies vascular inflammation and increases the risk of thrombosis in diabetic patients.
Recognising this depletion raises an immediate question for glutathione in Type 2 Diabetes interventions.
Can standard oral GSH actually restore what diabetes destroys?
Oral GSH vs Liposomal Glutathione: The Bioavailability Gap
Standard oral supplements fail to raise blood levels because stomach enzymes break the molecule apart before it is absorbed. Advanced liposomal technology protects the nutrient within a lipid shell, ensuring it reaches the bloodstream intact.
Here is a tabular representation to understand the difference between oral and liposomal glutathione:
| Parameter | Standard Oral GSH | Liposomal Glutathione |
| GI Stability | Intestinal GGT and proteases in the jejunum cleave the tripeptide; 80% or more undergo degradation. | Phospholipid bilayers physically shield the molecule; 70% plus remains intact through the gut. |
| Peak Plasma (Cmax) | Multiple clinical trials show no rise in blood levels after a 1000 mg oral dose. | The form achieves 6 times higher peak plasma levels at equivalent doses [5]. |
| Absorption Path | The gut absorbs only free amino acids via passive diffusion after total tripeptide cleavage. | Cells use endocytosis and membrane fusion to transport intact liposomes into the cell. |
| Cellular Delivery | Cells must synthesise new GSH from fragments; this process fails in diabetic states. | Liposomes deposit GSH directly into the cytoplasm; PBMC levels rise by 100% [6]. |
| Plasma Retention | The body clears these fragments quickly; no elevation persists beyond 3 hours. | A slow-release profile keeps plasma levels high for 24 hours post-dose. |
| First-Pass Loss | The liver extracts a high percentage of the absorbed amino acid fractions. | Encapsulation bypasses the portal path via lymphatic uptake to boost reach. |
Clinical trials on liposomal glutathione show a 35% reduction in 8-isoprostane and a 20% improvement in the GSSG:GSH ratio. NK cell activity rises by 400% within 14 days, while lymphocyte proliferation gains a 60% boost [7]. Research also notes increased IFN-γ and IL-2 with reduced IL-6 and IL-10 levels.
These results confirm the high bioavailability of liposomal glutathione for systemic health. This form bypasses first-pass metabolism to provide effective Glutathione support for Type 2 Diabetes.
Also read: Your Top 5 Questions About Liposomal Glutathione
Liposomal Glutathione and Cellular Immunity: Clinical Evidence
Clinical trials confirm that liposomal glutathione restores immune function in diabetic patients within 3 months, whereas standard interventions consistently fall short [8]. It directly targets GSH depletion in hyperglycemia, producing outcomes measurable at the cellular immune level.
- Immune Response: Patients who supplement with liposomal GSH show measurable immune improvements within just 2 weeks of starting supplementation. These rapid gains highlight how starved immune cells are of glutathione for Type 2 Diabetes recovery.
- Stronger Infection Defence: Study participants show improved ability to fight intracellular bacterial infections following liposomal GSH supplementation. The outcome confirms that restored GSH levels translate into real-world immune competence, not just improved lab markers.
- Lower Inflammatory Load: Supplemented patients recorded significant drops in pro-inflammatory cytokines that sustain chronic immune exhaustion in diabetic subjects. Correcting GSH depletion in hyperglycemia breaks the inflammatory cycle that standard diabetes treatments rarely address directly.
- API Quality Matters: Clinical outcomes from trials are only reproducible when the liposomal GSH API maintains verified encapsulation efficiency and a stable GSH: GSSG ratio. Poor API quality silently undermines product efficacy regardless of the supporting clinical evidence behind the ingredient.
Clinical efficacy, however, is only as reliable as the manufacturing process that produces the liposomal GSH API behind every finished formulation.
Manufacturing Stable Liposomal Glutathione APIs for Nutraceuticals
Producing pharmaceutical-grade glutathione for Type 2 Diabetes requires precise control over chemical and physical parameters to ensure the final product delivers real clinical value.
- Lipid Selection and Matrix Composition: High-purity phosphatidylcholine from non-GMO sunflower sources serves as the essential backbone of the phospholipid matrix, ensuring stable membrane fluidity and high encapsulation efficiency. Manufacturers frequently incorporate cholesterol at a 7:3 molar ratio to increase membrane rigidity and prevent premature leakage of the active tripeptide during storage.
- Critical Quality Attributes (CQAs): The target particle size for optimal absorption must stay between 80nm and 200nm to ensure the vesicles move through the gut wall effectively. Technical teams maintain a zeta potential of 30mVs or higher to prevent particle aggregation and ensure the suspension remains stable throughout the entire distribution process for liposomal products.
- Encapsulation and Redox Ratio Control: A commercially viable API must achieve an encapsulation efficiency of 70% or higher to justify the supplement’s cost and performance. Production takes place under a strict inert atmosphere, using nitrogen or argon to prevent oxidation of reduced GSH to its inactive GSSG form.
- Advanced Size Reduction Technologies: High-pressure homogenisation and microfluidisation enable the large-scale production of liposomes while maintaining a very tight, consistent particle size distribution across every batch. Tangential flow filtration serves as a final step for a CDMO to remove unencapsulated material and concentrate the potent fraction for this application and technology.
- Stability and Format Strategy: Lyophilisation or freeze-drying with the help of cryoprotectants like trehalose converts the liquid suspension into a stable powder for use in capsules or tablets. This process extends the shelf life to 36 months and protects the delicate phospholipid structure from the risks of peroxidation or particle growth over time.
- GMP Compliance and Quality Assurance: A manufacturer must provide a full Certificate of Analysis that covers heavy metal limits and microbial counts to meet international regulatory standards. Manufacturers must hold valid GMP certifications to prove that every batch meets the high safety requirements for products sold in India, the US, or Europe.
WBCIL’s liposomal glutathione API has been developed through iterative formulation refinement, achieving consistent particle size distribution within the 80-200 nm range and encapsulation efficiency above 70% across validated commercial-scale batches.
It provides these high-tier manufacturing standards to ensure global pharmaceutical and nutraceutical partners receive a stable, clinically validated API for their specialised formulations.
Final Thoughts
Blood sugar management alone fails to restore the immune health that glutathione for Type 2 Diabetes directly protects. Peer-reviewed clinical trials now validate a specific ingredient strategy for formulators and manufacturing partners. High-tier supplements must feature verified encapsulation efficiency and confirmed redox ratios before a brand commits to a formula. Sourcing from manufacturers like WBCIL removes the quality risks that often undermine specialised nutraceutical development. The advanced approach ensures the final product delivers the true biological impact demanded by current science.
- Sekhar, R.V., McKay, S.V., Patel, S.G., Guthikonda, A.P., Reddy, V.T., Balasubramanyam, A. and Jahoor, F. (2010). Glutathione Synthesis Is Diminished in Patients With Uncontrolled Diabetes and Restored by Dietary Supplementation With Cysteine and Glycine. Diabetes Care, [online] 34(1), pp.162–167.
- Lutchmansingh, F.K., Hsu, J.W., Bennett, F.I., Badaloo, A.V., McFarlane-Anderson, N., Gordon-Strachan, G.M., Wright-Pascoe, R.A., Jahoor, F. and Boyne, M.S. (2018). Glutathione metabolism in type 2 diabetes and its relationship with microvascular complications and glycemia. PLOS ONE, [online] 13(6), p.e0198626.
- Daryabor, G., Atashzar, M.R., Kabelitz, D., Meri, S. and Kalantar, K. (2020). The Effects of Type 2 Diabetes Mellitus on Organ Metabolism and the Immune System. Frontiers in Immunology, 11(1582).
- Berbudi, A., Khairani, S. and Tjahjadi, A. (2025). Interplay Between Insulin Resistance and Immune Dysregulation in Type 2 Diabetes Mellitus: Implications for Therapeutic Interventions. ImmunoTargets and Therapy, Volume 14, pp.359–382.
- Prasad, K.N., C, C., Karthik, Y., Girish, G.V. and Phadnis, S. (2026). Liposomal Glutathione Outperforms Plain Glutathione in Uptake, Cell Regeneration, and Systemic Availability: Evidence from Cellular and Human Models. British Journal of Nutrition, pp.1–24.
- Sinha, R., Sinha, I., Calcagnotto, A., Trushin, N., Haley, J.S., Schell, T.D. and Richie, J.P. (2018). Oral supplementation with liposomal glutathione elevates body stores of glutathione and markers of immune function. European Journal of Clinical Nutrition, [online] 72(1), pp.105–111.
- Sinha, R., Sinha, I., Calcagnotto, A., Trushin, N., Haley, J.S., Schell, T.D. and Richie, J.P. (2018). Oral supplementation with liposomal glutathione elevates body stores of glutathione and markers of immune function. European Journal of Clinical Nutrition, [online] 72(1), pp.105–111.
- To, K., Cao, R., Yegiazaryan, A., Owens, J., Nguyen, T., Sasaninia, K., Vaughn, C., Singh, M., Truong, E., Medina, A., Avitia, E., Villegas, J., Pham, C., Sathananthan, A. and Venketaraman, V. (2021). Effects of Oral Liposomal Glutathione in Altering the Immune Responses Against Mycobacterium tuberculosis and the Mycobacterium bovis BCG Strain in Individuals With Type 2 Diabetes. Frontiers in Cellular and Infection Microbiology, 11.
Glutathione neutralises free radicals by donating electrons to convert them into stable, harmless forms. It also suppresses inflammatory signals, reducing both immune dysfunction and insulin resistance. Restored glutathione levels in Type 2 Diabetes ensure cells remain protected from ongoing internal damage.
High blood sugar diverts the fuel needed to recycle this antioxidant, slowing the activity of the enzymes that produce it. The process also creates harmful compounds that bond to and drain existing stores. The overall result is a progressive deficiency that worsens as the condition develops.
A phospholipid bilayer protects the nutrient from gut enzymes that usually break it apart. This shell allows the molecule to enter the bloodstream directly and fuse with cell membranes. This method deposits the active tripeptide into cells where standard oral powders fail.
The ingredient works well in softgels, liquid sachets, and effervescent tablets for various health products. It carries a safe status and simplifies the regulatory process for new product launches. Formulators must verify stable particle size and high encapsulation efficiency to achieve optimal results.
A trusted supplier must provide full GMP compliance and detailed data on particle size and purity. WBCIL manufactures high-grade liposomal glutathione APIs with the technical documentation required for global pharmaceutical and nutraceutical standards. Their focus on batch consistency helps partners build products that match clinical claims.











