Ubiquinol vs Liposomal CoQ10: A Formulator’s Guide
The evaluation of CoQ10 formulations requires a clear understanding of both molecular properties and delivery limitations. You may already recognise how inconsistent CoQ10 performance can be across products, even when dosing appears appropriate. The comparison of ubiquinol vs. liposomal CoQ10 often focuses on redox form. This overlooks the core limitation of absorption. CoQ10 has a high molecular weight and a crystalline structure. It also shows poor water solubility, which limits dispersion and uptake.
Before you compare forms, you need to understand how delivery architecture determines absorption efficiency and systemic availability.
Key Takeaways:
- CoQ10 bioavailability depends more on dispersion and delivery system design than redox form selection across nutraceutical formulations.
- WBCIL’s Lipoedge TM liposomal encapsulation improves stability, protects CoQ10 during digestion, and enables consistent cellular uptake beyond conventional absorption pathways.
- Ubiquinol may oxidise during gastrointestinal transit, which reduces its stability advantage under real formulation and storage conditions.
Quick Answer: Liposomal CoQ10 offers more reliable absorption and stability, while ubiquinol performance depends heavily on formulation conditions and gastrointestinal conversion.

Why CoQ10 Absorption Is a Formulation Problem
Before you can compare ubiquinol vs. liposomal CoQ10, you need to understand why CoQ10 is structurally difficult to absorb in any form. CoQ10’s large molecular weight, extreme lipophilicity, and crystalline solid state make it one of the most absorption-resistant compounds in oral supplement manufacturing
The following evidence shows that formulation architecture resolves this problem far more reliably than redox-form selection alone.
- CoQ10 has a molecular weight above 860 daltons and near-zero aqueous solubility, which severely limits its dispersion in the gastrointestinal tract. Oral bioavailability from a single unformulated dose has been reported as low as 2-3% in preclinical models. [1]
- Poor dispersion of CoQ10 crystals in the gut can reduce bioavailability by up to 75%. This applies to both ubiquinone and ubiquinol [2]. Crystal dispersion has a greater impact on plasma CoQ10 levels. This impact exceeds the effect of redox form selection.
- A six-week crossover study in healthy volunteers found that CoQ10 bioavailability varied significantly between individuals. The intrasubject variability means that delivery system consistency matters more than form at the population level.
- CoQ10 requires dietary fat and bile salts for absorption. Fasting conditions can reduce its uptake significantly. For softgel and functional beverage production, this fat-dependency directly shapes which delivery architectures are viable.
- A review found that most human studies on nanoemulsions and dispersions reported fractional absorption rates ranging from 1.5% to 12.5% [3]. Such conventional approaches are what position liposomal technology as a technically distinct solution.
The comparison of ubiquinol vs. liposomal CoQ10 starts with stability, absorption, and conversion differences.
| Parameter | Ubiquinone (Oxidised CoQ10) | Ubiquinol (Reduced CoQ10) |
| Plasma absorption evidence | A crossover RCT found no statistically significant bioavailability difference between ubiquinone and ubiquinol. | A separate study found ubiquinol raised plasma CoQ10 to nearly double the level of ubiquinone under softgel excipients [4]. |
| Gastrointestinal stability | Ubiquinone is stable; it does not oxidise and reaches intestinal absorption in its original state. | A study found that between 54-76% of ubiquinol oxidises to ubiquinone within 60 mins. in simulated gastric conditions [5]. |
| Manufacturing complexity and cost | Stable at room temperature; resistant to light and air exposure. | Must be encapsulated under nitrogen to prevent pre-fill oxidation; requires antioxidant stabilisers such as tocopherols. |
| Clinical evidence base | Ubiquinone carries a significantly larger body of cardiovascular and neurodegenerative RCT data, including the Q-SYMBIO trial on heart failure. [6] | A 2026 RCT found that a novel ubiquinol cocrystal formulation showed higher systemic bioavailability versus ubiquinone reference (AUC ratio 2.01) [7]. |
| Conversion in the body post-absorption | After intestinal absorption, ubiquinone enters chylomicrons and is reduced to ubiquinol; more than 90% of circulating CoQ10 appears as ubiquinol [8]. | Ubiquinol taken orally is largely oxidised to ubiquinone in the GI tract. |
Also read: Defining API Quality at WBCIL
How Liposomal CoQ10 Delivery Works
You need to understand how pharmaceutical-grade CoQ10 reaches cells. This explains why liposomal encapsulation improves absorption rates.
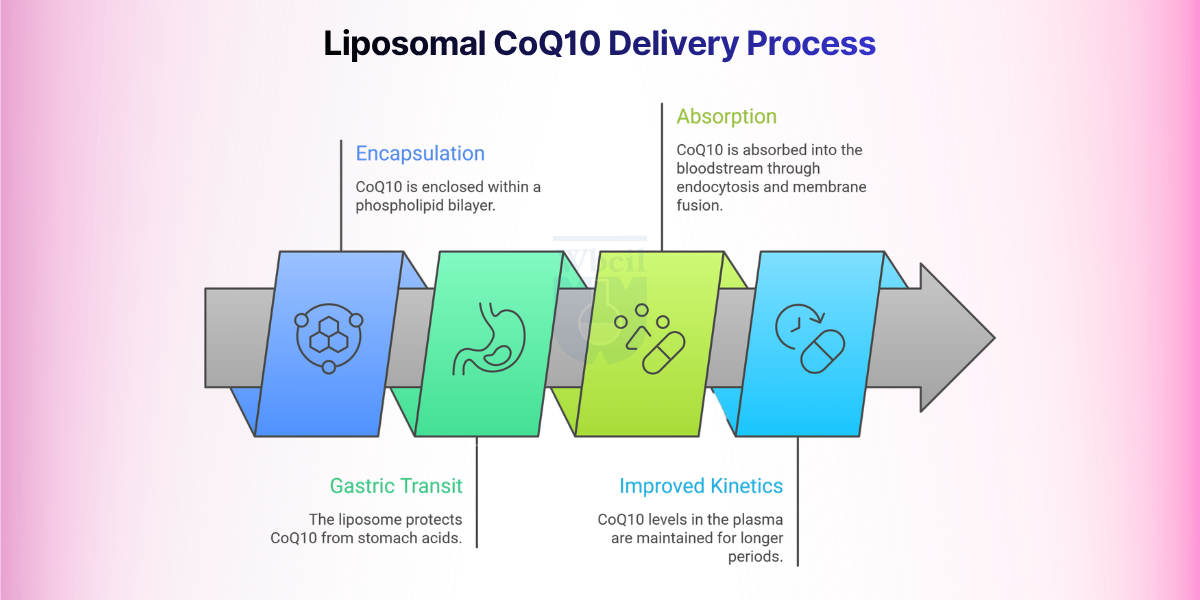
Here is a step-wise process for the liposomal CoQ10 delivery:
Step 1: Encapsulation Within a Phospholipid Bilayer
CoQ10 is embedded within a nanoscale phospholipid vesicle during manufacturing. The bilayer is composed of phosphatidylcholine from sunflower lecithin, which structurally mirrors human cell membranes. This architecture keeps CoQ10 in a pre-dispersed, amorphous state, eliminating the crystal dissolution problem that limits standard formulations.
Step 2: Protection Through Gastric Transit
The phospholipid shell resists breakdown by stomach acid, bile salts, and digestive enzymes. This protection is critical because unencapsulated CoQ10 loses significant potency before it reaches the small intestine. A sunflower lecithin-based liposomal formulation, used in a 2025 double-blind RCT, demonstrated enhanced gastrointestinal stability and reduced enzymatic degradation versus standard CoQ10 [9].
Step 3: Absorption via Endocytosis and Membrane Fusion
Unlike standard CoQ10, which depends on bile-mediated micelle formation, liposomal CoQ10 absorbs through endocytosis and direct membrane fusion. This bypasses dietary fat dependency and reduces fed-versus-fasted absorption variability.
Step 4: Improved Plasma Concentration Kinetics
A 2025 double-blind, placebo-controlled RCT registered with India’s Clinical Trials Registry confirmed liposomal CoQ10 produced significantly higher plasma concentrations than non-liposomal CoQ10. Peak plasma concentration was higher, and total absorption over 24 hours was measurably greater at equivalent doses.
Understanding absorption rates of CoQ10 helps you evaluate why liposomal delivery performs more reliably across diverse nutraceutical formulation formats.
Liposomal CoQ10 in Nutraceutical Formulations
Formulating with pharmaceutical-grade coenzyme Q10 requires an API that performs across formats. Liposomal delivery opens product categories that conventional CoQ10 cannot reach.
Cardiovascular and Statin-Support Supplements
Statins inhibit the HMG-CoA reductase pathway, the same pathway responsible for synthesising endogenous CoQ10. This means statin user faces a dual burden: managed cholesterol but depleted CoQ10, which contributes to statin-associated muscle symptoms. Lipoedge TM Liposomal CoQ10 restores depleted levels more efficiently at a lower dose than conventional CoQ10 formulations. For formulators developing statin-support or cardiac health supplements, liposomal CoQ10 provides a clinically substantiated API with a verified safety profile.
Softgel and Capsule Production
Standard CoQ10 requires co-administration with dietary fat for meaningful absorption. Liposomal encapsulation makes CoQ10 water-dispersible, removing the fat-dependency that limits conventional softgel performance. A next-generation liposomal formulation in a 2026 crossover study achieved a 4.3-fold higher AUC versus conventional CoQ10 at equivalent doses. The dose efficiency directly reduces raw material cost per SKU, a commercially meaningful advantage for softgel manufacturers competing in a saturated CoQ10 category.
Functional Beverages and Powder Formulations
Conventional CoQ10 cannot function in water-based beverage matrices because of its crystalline, fat-soluble structure. WBCIL’s Lipoedge TM Liposomal CoQ10 powder disperses in water, oils, and dry mixes. Stability data on one liposomal CoQ10 formulation confirmed it retained over 98% potency in pH 3.0-7.0 beverages over twelve months with no sedimentation. Anti-inflammatory effects from CoQ10 supplementation are more pronounced with interventions lasting over ten weeks, so consistent plasma delivery across a beverage format is commercially significant. Liposomal CoQ10 in Anti-Ageing Cosmetics
Raw CoQ10 is structurally incompatible with most cosmetic matrices, its high molecular weight, extreme lipophilicity, and light sensitivity create penetration and stability problems that liposomal encapsulation directly resolves.
Why CoQ10 Depletes in Skin and Why It Matters
CoQ10 is present in all skin layers, but levels fall with age and UV exposure, reducing the skin’s capacity for cellular energy production and free radical defence. When CoQ10 is depleted, fibroblasts enter cellular senescence and reduce production of collagen and elastin. CoQ10 also reduces collagenase expression in dermal fibroblasts exposed to UV radiation, a direct mechanism for wrinkle prevention at the cellular level.
Liposomal Encapsulation Solves the Skin Penetration Problem
Unencapsulated CoQ10 sits on the skin surface; its large molecular weight and water insolubility prevent meaningful penetration into the dermis. Liposomes with diameters under 200 nm achieve this by fusing with epidermal cell membranes, releasing CoQ10 into deeper skin layers. A separate in vivo study using a protransfersomal CoQ10 emulgel confirmed increased collagen density and fibroblast count in UV-aged skin, with no erythema or skin irritation observed. For cosmeceutical formulators, this dual evidence base, penetration data and collagen response data support substantiated anti-ageing claims.
Stability Considerations for Cosmetic Formulation
CoQ10 degrades under light, heat, and oxidative conditions, and this instability is more acute in cosmetic matrices than in oral supplement formats. Liposomal encapsulation forms a protective barrier. It shields CoQ10 from oxidation, pH changes, and light exposure. For a cosmeceutical brand launching in India’s rapidly expanding personal care segment, these combined stability and efficacy data enable compliant product claims.
Pharmaceutical-grade Coenzyme Q10 selection determines formulation stability, absorption consistency, and overall performance across diverse nutraceutical and cosmeceutical applications.
Sourcing Pharmaceutical-Grade Liposomal CoQ10
- When you source a liposomal CoQ10 API, you are not just buying an ingredient, you are buying a delivery system backed by manufacturing precision.
- WBCIL’s Lipoedge TM liposomal CoQ10 achieves encapsulation efficiency above 80%, with particle sizes typically below 200 nm. Zeta potential is maintained above −30 mV, preventing vesicle aggregation and preserving colloidal stability throughout the supply chain.
- Over 64 years of pharmaceutical API manufacturing experience underpins every batch. For B2B buyers exporting to regulated markets, this reduces both audit burden and time-to-launch.
- WBCIL supplies liposomal CoQ10 as a liquid liposomal dispersion or a free-flowing, water-dispersible powder. The liquid format suits serums, creams, and cosmeceutical bases requiring direct incorporation.
- WBCIL manufactures liposomal CoQ10 under WHO-GMP, cGMP, ISO, HACCP, and FSSAI certifications at its Kolkata and Dahej facilities. This certification stack supports simultaneous regulatory submissions to CDSCO, FDA, EMA, and FSSAI, without reformulation per market.
- WBCIL holds 17 active patents covering its liposomal technology platforms, including a patented solvent-free manufacturing process. This process protects CoQ10 from oxidative degradation during encapsulation.
West Bengal Chemical Industries Limited supplies liposomal CoQ10 API for nutraceutical OEM production, private-label finished products, and bulk raw ingredient programmes. The company serves over 4,000 clients across 75-plus countries with flexible minimum order quantities.
Final Thoughts
A structured evaluation of CoQ10 formulations requires prioritising delivery efficiency over chemical form alone. The comparison of ubiquinol vs. liposomal CoQ10 shows that dispersion, stability, and uptake pathways determine real outcomes.
If you aim for consistent plasma levels, focus on delivery systems that minimise variability across fed and fasted states.
You should also assess stability data, encapsulation efficiency, and format compatibility before finalising your formulation strategy. WBCIL can support this process with documented systems and formulation-ready Lipoedge TM liposomal APIs, without requiring major redevelopment.
Note: This content is intended for B2B pharmaceutical and nutraceutical professionals and does not constitute medical advice.
- Li, Z. and Kopec, R.E. (2024). CoQ10 Bioaccessibility and Caco-2 Cell Uptake Improved with Novel Medium Chain Triglyceride Encapsulation. Food & Function.
- Mantle, D. and Dybring, A. (2020). Bioavailability of Coenzyme Q10: An Overview of the Absorption Process and Subsequent Metabolism. Antioxidants, 9(5), p.386.
- Barakat, A., Shegokar, R., Dittgen, M. and Müller, R.H. (2013). Coenzyme Q10 oral bioavailability: effect of formulation type. Journal of Pharmaceutical Investigation, 43(6), pp.431–451.
- Langsjoen, P.H. and Langsjoen, A.M. (2013). Comparison study of plasma coenzyme Q10levels in healthy subjects supplemented with ubiquinol versus ubiquinone. Clinical Pharmacology in Drug Development, 3(1), pp.13–17.
- Judy, W.V. (2021). The Instability of the Lipid-Soluble Antioxidant Ubiquinol: Part 1—Lab Studies. Integrative Medicine: A Clinician’s Journal, [online] 20(4), p.24.
- Judy, W.V. (2021). The Instability of the Lipid-Soluble Antioxidant Ubiquinol: Part 3–Misleading Marketing Claims. Integrative Medicine: A Clinician’s Journal, [online] 20(6), p.24.
- Mei, X., Zhu, B., Soni, K., Kasaraneni, K. and Panchal, N. (2026). A Randomized, Double‐Blind, Two‐Treatment, Two‐Period, Crossover Study Investigating the Systemic Bioavailability of a Novel Cocrystal Ubiquinol Formulation Compared with a Ubiquinone Formulation in Healthy Adults. Clinical Pharmacology in Drug Development, 15(3).
- Clinical Applications of Scientific Innovation (2026). Ubiquinone vs. Ubiquinol: Does the Form of CoQ10 Matter – Or Is Delivery the Real Key? [online] Designs for Health.
- Ralf Jäger, Purpura, M., Ashok Godavarthi, Ceylan, H.I., Balcombe, S.T., Ambrish Chandrappa and Tinsley, G.M. (2025). Impact of liposomal delivery on coenzyme Q10 absorption: a double-blind, placebo-controlled, randomized trial. Frontiers in Nutrition, 12, pp.1605033–1605033.
Ubiquinol is the reduced form of CoQ10, while liposomal CoQ10 refers to a delivery system that improves dispersion and absorption. Ubiquinol may offer higher initial bioavailability, but liposomal CoQ10 provides more consistent absorption by bypassing solubility limitations.
Liposomal CoQ10 often shows more reliable bioavailability because it improves dispersion and enables absorption through endocytosis. Ubiquinol performance depends on formulation stability and can vary due to oxidation during gastrointestinal transit.
Yes, liposomal CoQ10 is water-dispersible and remains stable across multiple formats, including beverages, powders, and capsules. This flexibility allows formulators to develop products without relying on dietary fat for absorption.
You should evaluate encapsulation efficiency, particle size, zeta potential, and stability data. These factors determine absorption performance, shelf life, and consistency across production batches.
CoQ10 has a high molecular weight, crystalline structure, and poor water solubility. These properties limit its dispersion in the gastrointestinal tract, which directly reduces absorption and systemic availability.