Cosmetics vs Cosmeceuticals: Know the Differences
You pick up a moisturiser, flip the bottle, and see a list of ingredients that reads like a chemistry textbook. But here is a question most people never ask — is this product actually doing something to your skin, or just sitting on top of it?
That one question is exactly where the difference between cosmetics and cosmeceuticals begins. And if you are a CDM/CMO buyer, a formulator, or simply someone who takes skincare seriously, understanding this distinction could change the way you source, manufacture, or choose products entirely.
Key Takeaways:
- The grade of your ingredients matters: A cosmeceutical formula built on cosmetic-grade raw materials will underperform. Pharmaceutical-grade Zinc, Magnesium, and other mineral salts are non-negotiable for high-performance functional skincare.
- Regulatory awareness is a competitive advantage: Understanding FDA vs EMA guidelines for cosmeceutical labelling helps brands and CDMOs navigate claims intelligently — protecting them legally while maximising product credibility.
- Cosmeceuticals are the future of skincare — but only when done right: The growing demand for bioactive, therapeutic-grade cosmetics is real. Brands that invest in proper sourcing, formulation science, and manufacturing standards are the ones that will earn lasting consumer trust.
What Are Cosmetics? A Simple Definition
Cosmetics are products designed to cleanse, beautify, or alter the appearance of the body without affecting its structure or function. Think of your daily face wash, lipstick, foundation, or body lotion. They work on the surface — and that is where their job ends.
Under the US FDA framework [1], a product is classified as a cosmetic if it does not claim to treat, prevent, or cure any condition. The European Medicines Agency (EMA) follows a similar logic under the EU Cosmetics Regulation (EC) No 1223/2009 [2], where cosmetics are defined strictly as substances applied to external body parts for aesthetic or hygiene purposes.
Common cosmetic ingredients include:
- Emollients and humectants (glycerin, shea butter)
- Surfactants for cleansing
- Fragrances and colorants
- Preservatives to extend shelf life
None of these change how your skin cells behave. They improve how your skin looks or feels — temporarily.

What Are Cosmeceuticals? Where Science Meets Skincare
Cosmeceuticals sit in a fascinating middle ground. The term itself is a blend of “cosmetics” and “pharmaceuticals,” and it describes products that contain bioactive skincare ingredients capable of influencing biological processes in the skin.
These are not just moisturisers. We are talking about formulations with retinoids, peptides, alpha hydroxy acids, niacinamide, and critically — pharmaceutical-grade mineral salts like Zinc and Magnesium — that interact with skin cells at a deeper level [3].
A cosmeceutical product might:
- Stimulate collagen synthesis
- Reduce melanin production to address pigmentation
- Regulate sebum output
- Strengthen the skin barrier at a cellular level
- Deliver active ingredients through advanced topical drug delivery systems
In short, cosmeceuticals do not just decorate the skin — they work with it.
That said, it is worth noting that the FDA does not officially recognise “cosmeceutical” as a legal category [1].
A product is either a cosmetic or a drug. However, the cosmeceutical segment has grown enormously in the industry, with manufacturers voluntarily holding their formulations to pharmaceutical-grade standards even when regulatory classification does not demand it.
Why Bioactive Skincare Ingredients Are the Heart of Cosmeceuticals?
The reason cosmeceuticals deliver visible, measurable results is largely down to the quality and concentration of their bioactive skincare ingredients. Unlike cosmetic-grade raw materials, bioactive ingredients are selected for their ability to trigger a biological response in the skin.
Some of the most widely used bioactives in cosmeceutical manufacturing include:
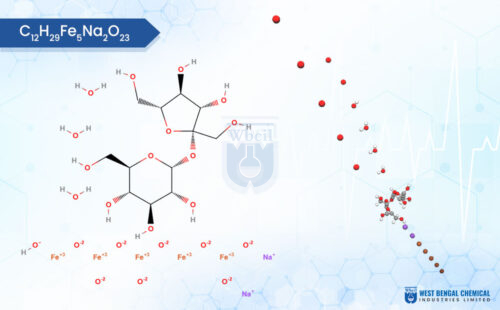
1. Mineral Salts — Zinc and Magnesium
Zinc is one of the most versatile minerals in functional skincare manufacturing. It plays a role in wound healing, sebum regulation, and the control of inflammation. Magnesium, on the other hand, supports enzymatic reactions that keep the skin barrier functioning well.
But here is the thing — not all Zinc or Magnesium is created equal. Using cosmetic-grade mineral salts in a cosmeceutical formulation is like building a precision instrument with everyday hardware. The purity, particle size, and solubility of pharmaceutical-grade Zinc and Magnesium directly impact how well these minerals penetrate and function in the skin [4].
This is why CDMOs and high-performance skincare brands specifically source pharmaceutical-grade Zinc and Magnesium for cosmetics — to ensure the ingredient actually does what the formula promises.
2. Peptides and Retinoids
Peptides are small chains of amino acids that signal skin cells to produce more collagen or elastin. Retinoids (derivatives of Vitamin A) are among the most clinically studied bioactives, proven to increase cell turnover and reduce fine lines. Both require careful formulation and stability management — something that goes far beyond standard cosmetic manufacturing.
3. Alpha Hydroxy Acids (AHAs) and Beta Hydroxy Acids (BHAs)
These exfoliating acids work by loosening the bonds between dead skin cells, encouraging faster renewal. At therapeutic concentrations, they cross into pharmaceutical-grade territory, which is why their percentage usage is regulated differently across markets.
Regulatory Differences Between Cosmetics and Cosmeceuticals
This is where things get particularly important for CDMOs and sourcing teams. The regulatory differences between cosmetics and cosmeceuticals vary significantly depending on the market.
FDA Guidelines (USA)
The FDA [1] does not recognise “cosmeceutical” as a product category. A product claiming to change the structure or function of the skin is legally a drug — and must go through the full drug approval process. This is why most manufacturers in the US carefully phrase product claims to stay within cosmetic territory, even when using pharmaceutical-grade APIs internally.
EMA Guidelines (EU)
The EMA and the EU Cosmetics Regulation [2] take a similar binary view. However, the EU framework is arguably stricter on ingredient safety assessments, required stability testing, and Product Information Files (PIF). Brands operating across EU markets need to ensure their cosmeceutical-grade products meet these documentation requirements even if the product is filed as a cosmetic.
What This Means for Manufacturers?
For CDMOs, the challenge is manufacturing to pharmaceutical standards while labelling to cosmetic regulations. This dual compliance requirement is one of the core manufacturing challenges of active ingredient skincare. It demands:
- Validated sourcing of pharmaceutical-grade APIs
- Robust stability and compatibility testing
- Precise documentation for regulatory submissions
- Expertise in topical drug delivery systems that ensure bioavailability without exceeding cosmetic claim boundaries

Why Mineral-Based Cosmeceuticals Are Gaining Ground?
There is a growing movement within functional skincare manufacturing toward mineral-based cosmeceuticals. And the reason is straightforward — minerals like Zinc, Magnesium, Copper, and Iron are naturally occurring, well-tolerated, and increasingly backed by clinical research.
When sourced at pharmaceutical grade, these minerals offer:
- High bioavailability for deeper skin penetration
- Compatibility with sensitive skin formulations
- Stability that synthetic actives often lack
- Clean label appeal for brands targeting health-conscious consumers
For CDMO buyers sourcing pharmaceutical-grade APIs for cosmeceutical formulations, this shift means partnering with manufacturers who understand both the chemistry and the compliance side.
WBCIL (West Bengal Chemical Industries Limited) has been supplying pharmaceutical-grade mineral salts — including Zinc, Magnesium, Calcium, and Iron — to skincare and nutraceutical manufacturers globally. Their product range, built to strict GMP standards, makes them a reliable source for cosmeceutical-grade raw materials.
Learn more at www.wbcil.com.
Manufacturing Challenges of Active Ingredient Skincare
Making a cosmeceutical is not the same as making a cosmetic. The formulation complexity, quality requirements, and regulatory scrutiny are all higher. Here are some of the key challenges manufacturers face:
Stability of Active Ingredients
Bioactives like retinol, Vitamin C, and peptides are notoriously unstable. They degrade when exposed to light, heat, or air. Packaging, pH management, and encapsulation technology — including liposomal delivery systems — are essential tools in maintaining efficacy.
Topical Drug Delivery Systems
Getting an active ingredient to where it needs to work in the skin requires more than just dissolving it in a cream base. Technologies like liposomes, nanoparticles, and microemulsions are increasingly used in cosmeceutical manufacturing to enhance penetration and controlled release [5].
Ingredient Sourcing and Purity
Perhaps the most underrated challenge. A cosmeceutical formula is only as good as its raw materials. Sourcing pharmaceutical-grade APIs — rather than cosmetic-grade alternatives — can be the difference between a product that delivers results and one that merely claims to.
- U.S. Food and Drug Administration (FDA). “Is It a Cosmetic, a Drug, or Both? (Or Is It Soap?)” — FDA.gov. Accessed 2025.
- European Commission. Regulation (EC) No 1223/2009 of the European Parliament and of the Council on cosmetic products. Official Journal of the European Union, 2009.
- Draelos, Z.D. (2009). Cosmeceuticals: Undefined, Unclassified, and Unregulated. Clinics in Dermatology, 27(5), 431–434.
- Gupta, M., et al. (2014). Zinc therapy in dermatology: A review. Dermatology Research and Practice, 2014, 709152.
- Benson, H.A.E. (2005). Transdermal drug delivery: Penetration enhancement techniques. Current Drug Delivery, 2(1), 23–33.
Cosmetics work on the surface of the skin for aesthetic purposes — they do not change the skin’s structure or function. Cosmeceuticals contain bioactive ingredients that interact with skin biology at a deeper level, offering functional benefits like collagen stimulation, pigmentation reduction, or barrier repair.
In the US, the FDA does not officially recognise “cosmeceutical” as a legal product category — a product is either a cosmetic or a drug. In the EU, the EMA follows a similar binary framework. However, many manufacturers voluntarily apply pharmaceutical manufacturing standards to cosmeceutical products, even when they are filed as cosmetics.
Zinc plays a key role in sebum regulation, wound healing, and inflammation control. Pharmaceutical-grade Zinc salts offer higher purity and bioavailability compared to cosmetic-grade alternatives, meaning they can actually deliver these benefits within a skincare formula.
Bioactive skincare ingredients are compounds that trigger a measurable biological response in the skin. Examples include retinoids (Vitamin A derivatives), peptides, niacinamide, AHAs/BHAs, and pharmaceutical-grade mineral salts like Zinc and Magnesium.
Common delivery systems include liposomes, microemulsions, nanoparticles, and encapsulation technology. These help stabilise active ingredients and improve penetration into the deeper layers of the skin where they can produce the intended effect.
Reputable API and mineral salt manufacturers like WBCIL (West Bengal Chemical Industries Limited) supply pharmaceutical-grade Zinc, Magnesium, Iron, Calcium, and other minerals to cosmeceutical and nutraceutical manufacturers worldwide. Visit www.wbcil.com for product details and sample requests.