Formulating Calcium Citrate for Gastric-Sensitive Patients
Calcium deficiency in gastric-sensitive patients is not simply a dietary problem; it is a physiological one rooted in how the stomach processes supplements. Conditions such as achlorhydria, chronic PPI use, and post-bariatric surgery all compromise the gastric environment that most calcium salts depend on for absorption.
The clinical and formulation consequences of choosing the wrong calcium salt are measurable: elevated parathyroid hormone levels, accelerated bone resorption, and increased fracture risk. Pharmaceutical manufacturers working with these patient populations carry a direct responsibility to get this ingredient decision right, from API specification through to finished dosage form.
In this blog, you will find a complete clinical and formulation guide covering why calcium citrate outperforms carbonate in gastric-sensitive patients and how to source high-purity mineral salts.
Key Takeaways:
- Calcium citrate absorbs independently of gastric acid; it remains the only reliable calcium salt for achlorhydric, PPI-dependent, and post-bariatric patients.
- Post-bariatric patients require 1,200-1,500 mg of elemental calcium daily, with no more than 500-600 mg per serving.
- Co-formulating calcium citrate with vitamin D3 and magnesium citrate completes the full absorption chain that calcium alone cannot deliver.
Quick Answer: Calcium citrate is the preferred pharmaceutical calcium salt because it dissolves and absorbs reliably without dependence on gastric acid. This makes it clinically superior to calcium carbonate for achlorhydric, post-bariatric, and PPI-dependent patient populations.

Why Gastric Physiology Drives Calcium Salt Selection
Calcium absorption begins in the stomach, where gastric acid ionises calcium salts into their absorbable elemental form. In gastric-sensitive patients, this process fails, and calcium citrate is the only clinically sound formulation option.
Here are some of the primary factors which contribute towards calcium salt selection:
- Gastric acid ionises calcium salts before intestinal absorption can occur. Calcium carbonate requires a gastric pH of 2-3 to dissolve adequately for absorption [1]. Without sufficient acid, the body absorbs far less calcium than the label dose indicates.
- PPI therapy raises intragastric pH to levels where carbonate cannot dissolve. Omeprazole and pantoprazole suppress acid secretion, raising gastric pH to 5-7. Calcium citrate absorbs reliably regardless of this pH shift.
- Bariatric surgery reduces both acid output and duodenal absorptive surface. A Roux-en-Y bypass reroutes food away from the primary site of calcium absorption. ASMBS guidelines specify calcium citrate as the preferred post-surgical supplement.
- Atrophic gastritis from pylori destroys acid-secreting parietal cells progressively. H. pylori prevalence is notably high across South Asian populations, including India. This makes calcium citrate a priority formulation for Indian manufacturers.
- Persistent calcium malabsorption triggers secondary hyperparathyroidism and bone resorption. Parathyroid hormone rises to compensate, pulling calcium directly from bone tissue [2]. Formulators who choose calcium citrate prevent this documented pathological cascade.
Also read: Calcium Citrate Malate vs. Calcium Dobesilate: Which Supports Better Vascular and Bone Health?
Calcium Citrate vs. Carbonate: The Absorption Evidence
Choosing between calcium salts is not a matter of preference; it is a decision grounded in solubility chemistry and patient physiology. In gastric-sensitive populations, the biochemical differences between these two salts result in measurable differences in clinical outcomes.
Here is a tabular representation to understand the difference between calcium citrate and calcium carbonate
| Parameter | Calcium Citrate | Calcium Carbonate |
| Acid Dependency for Absorption | Absorbs independently of gastric pH, making it reliable across all patient populations, including those with achlorhydria or PPI use | Requires gastric pH of 2-3 for adequate dissolution; absorption drops significantly in acid-suppressed or fasted patients |
| Bioavailability Advantage | Absorbed 22-27% more effectively than carbonate in acid-compromised conditions, per the Sakhaee et al. meta-analysis of 15 clinical studies | Bioavailability is adequate only when gastric acid is present, and the supplement is taken with a meal |
| Gastrointestinal Tolerability | Well tolerated in sensitive stomachs; does not produce CO₂ during dissolution, so bloating, flatulence, and constipation remain low | Reacts with HCl to release CO₂ gas, directly causing flatulence, bloating, and constipation — particularly problematic in post-bariatric patients |
| Dosing Flexibility | Can be taken with or without food, giving formulators and patients greater compliance flexibility across clinical settings | Must be taken with meals to leverage food-stimulated acid secretion; fasted dosing produces unreliable absorption |
| Renal Stone Risk | Urinary citrate elevation from citrate salt inhibits calcium oxalate crystal formation, reducing kidney stone risk in susceptible patients | Does not confer this protective effect; high-dose carbonate supplementation may slightly increase urinary calcium without the citrate buffer |
Bariatric surgery creates a gastrointestinal environment where calcium carbonate simply cannot function, making calcium citrate stands as the only formulation-ready option.
Formulating Calcium Citrate for Bariatric Patients
Bariatric surgery permanently alters the digestive tract, and the ability to absorb calcium drops sharply after the procedure. Knowing exactly how these anatomical changes affect absorption determines how you correctly specify calcium citrate for gastric patients in oral dosage forms.
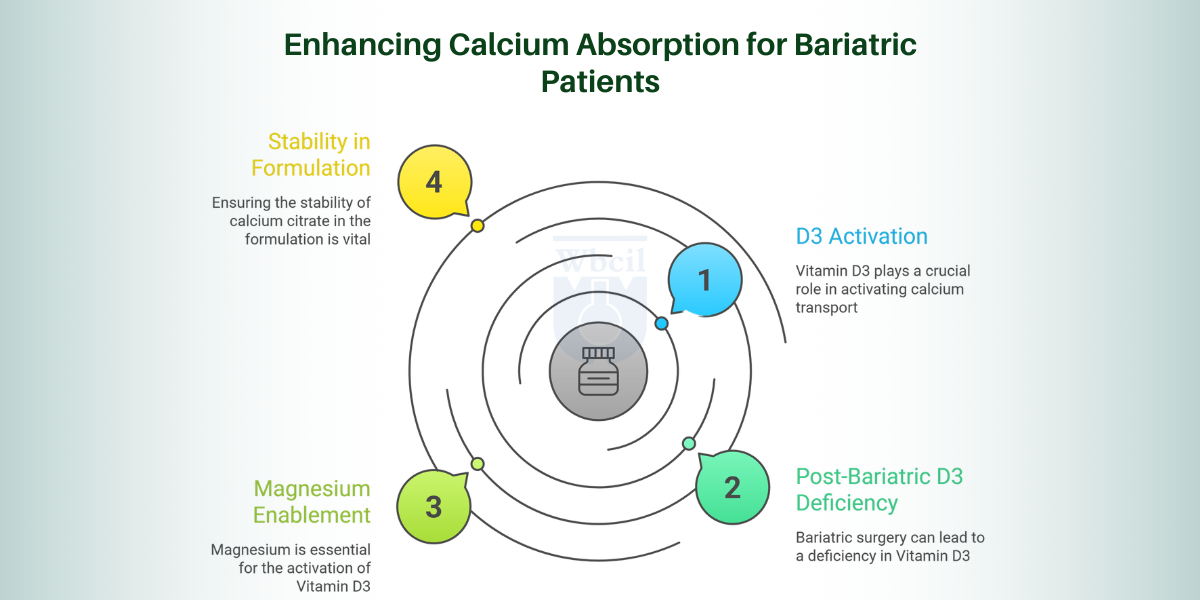
D3 Activates Transport
Vitamin D3 first enters the liver, where it is converted to 25-hydroxyvitamin D, then travels to the kidneys for final activation into calcitriol. Calcitriol directly stimulates calbindin-D9k, the intestinal protein that physically carries calcium across the gut wall. Without adequate calcitriol, calcium citrate reaches the intestine but cannot cross into systemic circulation efficiently [3].
Post-Bariatric D3 Deficiency
Fat-soluble vitamin absorption drops sharply after bariatric surgery because bile acid mixing, essential for fat-soluble nutrient uptake, reduces significantly. Patients with gastric sensitivity frequently present with pre-existing vitamin D insufficiency before surgery, which the procedure further compounds. Formulations that deliver calcium citrate without co-supplementing vitamin D3 address only half the clinical problem these patients face.
Magnesium Enables Activation
Magnesium acts as a cofactor for enzymes responsible for both hepatic and renal hydroxylation steps in vitamin D activation. Without sufficient magnesium, vitamin D3 supplementation cannot fully convert to calcitriol, leaving the calcium transport pathway incomplete regardless of the calcium citrate dose. This dependency means magnesium deficiency, common in post-bariatric and PPI-dependent patients, undermines the entire supplementation regimen.
Stability in Formulation
Vitamin D3 degrades with exposure to light, moisture, and elevated temperatures, requiring opaque packaging and controlled humidity during tabletting. Calcium citrate’s slightly alkaline microenvironment can accelerate D3 degradation in the absence of a protective excipient, such as microcrystalline cellulose or starch. Selecting a water-miscible cholecalciferol form and maintaining blend moisture below 3% preserve D3 potency through the intended shelf life.
Calcium citrate for gastric patients delivers its full clinical value only when vitamin D3 and magnesium are part of the formulation.
Vitamin D3 and Magnesium in Gastric-Sensitive Formulas
Calcium citrate for gastric patients is most effective when formulated with vitamin D3 and magnesium, as these co-nutrients complete the absorption pathway that calcium alone cannot complete.
- D3 Drives Absorption: Without calcitriol activating calbindin-D9k, calcium citrate is absorbed in the intestine but never reaches systemic circulation at therapeutic levels. Research shows that vitamin D supplementation increases fractional calcium absorption by approximately 65% in individuals with vitamin D deficiency.
- Surgery Deepens Deficiency. Studies show that up to 60% of bariatric surgery candidates present with vitamin D insufficiency before the procedure. Reduced bile acid mixing after bypass then compounds this deficit, making D3 co-formulation a clinical necessity.
- Magnesium Enables Activation: Both hepatic and renal hydroxylation of D3 to calcitriol are magnesium-dependent enzymatic reactions that cannot proceed without it. A 2013 Magnesium Research study found magnesium supplementation raised serum 25-hydroxyvitamin D levels by 30%.
- Three-Way Formulation Wins: This combination addresses calcium solubility, intestinal transport activation, and enzymatic D3 conversion within a single product. The Sakhaee et al. meta-analysis confirmed calcium citrate paired with D3 produced superior bone mineral density outcomes. (Bariatric Fusion)
- Stability Needs Attention: Calcium citrate’s alkaline microenvironment accelerates D3 degradation without a protective barrier such as microcrystalline cellulose. Studies confirm that D3 loses up to 40% of its potency within 6 months without adequate moisture and light protection.
Also read: Calcium Bisglycinate: Premier Choice for Gummies and Liquids
Sourcing USP-Grade Calcium Citrate API Through WBCIL
When the clinical case for calcium citrate for gastric patients is this well-established, the API quality determines whether your finished formulation delivers on that promise. WBCIL has over 64 years of experience in manufacturing pharmaceutical-grade mineral salts, certified under WHO-GMP, cGMP, and ISO 9001:2015 across facilities in Kolkata and Dahej, Gujarat.
Now, let’s understand why sourcing USP-grade calcium citrate is essential for B2B organisations:
- Pharmacopoeia-Grade Purity: WBCIL’s calcium citrate meets USP and IP standards, with 21% elemental calcium and a minimum 97.5% purity on a dried basis. Every batch undergoes heavy metal testing, microbial assessment, and particle size verification before release.
- Multiple Physical Forms: WBCIL supplies calcium citrate in ultrafine powder, granular, and pelletised forms for different manufacturing processes. This flexibility means your dosage form decision drives the API selection, not the other way around.
- Regulatory Documentation Ready: WBCIL maintains validated Drug Master Files in CTD format, with USFDA DMF submissions already completed. Your regulatory team receives audit-ready documentation for both domestic and export market filings in India.
- Co-Nutrient Premix Capability: WBCIL manufactures premix formulations combining calcium citrate with vitamin D3 and magnesium citrate in a single blend. This reduces your vendor qualification burden while delivering a clinically complete ingredient from one qualified source.
- Proven Export Track Record: WBCIL has consistently supplied calcium citrate API to manufacturers across Japan, Germany, the UK, Australia, and the MENA region. For manufacturers with international market ambitions, this export compliance history reduces supply chain quality risk.
Final Thoughts
Gastric sensitivity changes everything about how the body processes calcium, and your formulation must account for that from the ingredient stage onwards. Selecting calcium citrate for gastric patients is the first correct decision, but pairing it with vitamin D3 and magnesium citrate makes it clinically complete. Keep individual doses within 500-600 mg of elemental calcium to respect intestinal transport saturation limits, and choose your dosage form, chewable, film-coated, or sachet, based on the specific patient population your product targets.
On the sourcing side, insist on USP or IP pharmacopoeia-grade API with validated DMF documentation, verified heavy metal limits, and a supplier whose physical form range matches your manufacturing process.
Working with WBCIL, a WHO-GMP- and cGMP-certified bulk calcium citrate API supplier with over 64 years of experience in high-purity mineral salts, gives your formulation the quality foundation it needs to deliver on its clinical promise.
- Fritz, K., Taylor, K. and Parmar, M. (2023). Calcium carbonate. [online] PubMed.
- Clinical Trials.gov (2026). Efficacy of Calcium Citrate Versus Calcium Carbonate for the Management of Chronic Hypoparathyroidism. [online] Clinicaltrials.gov.
- California, M.S., PharmD, PhD Department of Pharmacy, Alta Bates Summit Medical Center Berkeley (2015). Pros and Cons of Calcium Supplements. [online]
Calcium citrate dissolves without gastric acid, making it reliably absorbable in patients with acid-compromised gastric conditions. Calcium carbonate requires a gastric pH of 2-3 for optimal absorption; without sufficient acid, absorption is reduced.
Select a soluble calcium citrate form that dissolves at near-neutral pH without acid assistance. Chewable tablets or powder sachets eliminate the need for gastric disintegration for these patients.
The Sakhaee et al. meta-analysis confirmed that calcium citrate absorbs 22-27% more effectively than calcium carbonate under acid-compromised conditions. ASMBS guidelines specifically recommend calcium citrate for post-bariatric patients with structurally reduced gastric acid output.
Verify purity meets the USP monograph minimum of 97.5% and confirm validated Drug Master Files in CTD format. Physical form availability, ultrafine powder, granular, or pelletised, should match your specific manufacturing process.
Calcium citrate dissociates across a wide pH range, performing consistently in low-acid gastric environments without food dependence. It offers superior GI tolerability and reduces kidney stone risk by elevating urinary citrate.