Evaluating Iron Polymaltose API Purity: A Buyer’s Guide
Iron deficiency anaemia affects over 1.2 billion people globally, and the quality of the API used in every iron formulation has direct consequences for patient outcomes [1]. For pharmaceutical manufacturers, sourcing the right iron preparation is not simply a procurement decision; it is a clinical responsibility. Iron hydroxide polymaltose purity sits at the centre of that responsibility, as batch inconsistencies in this structurally complex API translate directly into sub-therapeutic products reaching vulnerable patient populations.
Unlike simple iron salts, Iron Polymaltose Complexes (IPCs) have a macromolecular structure, so standard assay panels alone cannot confirm pharmaceutical-grade quality. A reality that catches many generic manufacturers unprepared at the regulatory submission stage.
In this blog, you will find a complete technical guide covering IPC purity parameters, molecular weight distribution, safety profile comparisons, and how to source a verified bulk iron polymaltose API that meets regulated market standards.
Key Takeaways:
- Iron hydroxide polymaltose purity above 98% with iron content exceeding 30% w/w is the pharmaceutical-grade benchmark for oral dosage forms.
- Molecular weight distribution, not iron content alone, determines therapeutic equivalence for generic IPC submissions in regulated markets.
- IPC’s non-ionic structure delivers measurably lower GI adverse events and broader formulation scope than ionic iron sucrose preparations.
Quick Answer: Iron hydroxide polymaltose purity is assessed through iron content above 30% w/w, minimum 98% purity, heavy metal limits, particle size below 50 microns, and free ionic iron control.

What Makes Iron Polymaltose a Complex API to Evaluate
The purity of iron hydroxide polymaltose cannot be assessed using a single test parameter. It requires a multi-attribute evaluation framework that accounts for the API’s macromolecular, heterogeneous structure.
Unlike simple iron salts, IPC is not a homo-molecular compound, and that structural complexity directly affects how you specify, test, and source it.
- Iron Polymaltose Complex (IPC) consists of an Fe³⁺ hydroxide core stabilised by a branched polymaltose carbohydrate shell, structurally comparable to ferritin. No two manufacturing processes produce an identical molecular distribution, making supplier selection a critical quality decision.
- IPC’s non-ionic character means it does not interact with food, medications, or gastric contents during oral administration. This property directly supports patient compliance and broadens your formulation brief.
- Research confirms that follow-on IPCs show measurable physicochemical variation from the originator product. Buyers sourcing for generic submissions must request independent characterisation data before qualification.
- Proprietary manufacturing protocols produce IPC batches with differing molecular weight distributions and colloidal particle sizes. Without standardised regulatory monographs, quality verification rests entirely with the buyer.
- Iron content percentage alone does not confirm therapeutic-grade IPC quality. Free ionic iron levels, particle size distribution, and colloidal stability are equally critical parameters that standard assay panels often miss.
Once you understand the complexity of IPC, the next step is to focus on the specific quality parameters that define pharmaceutical-grade material.
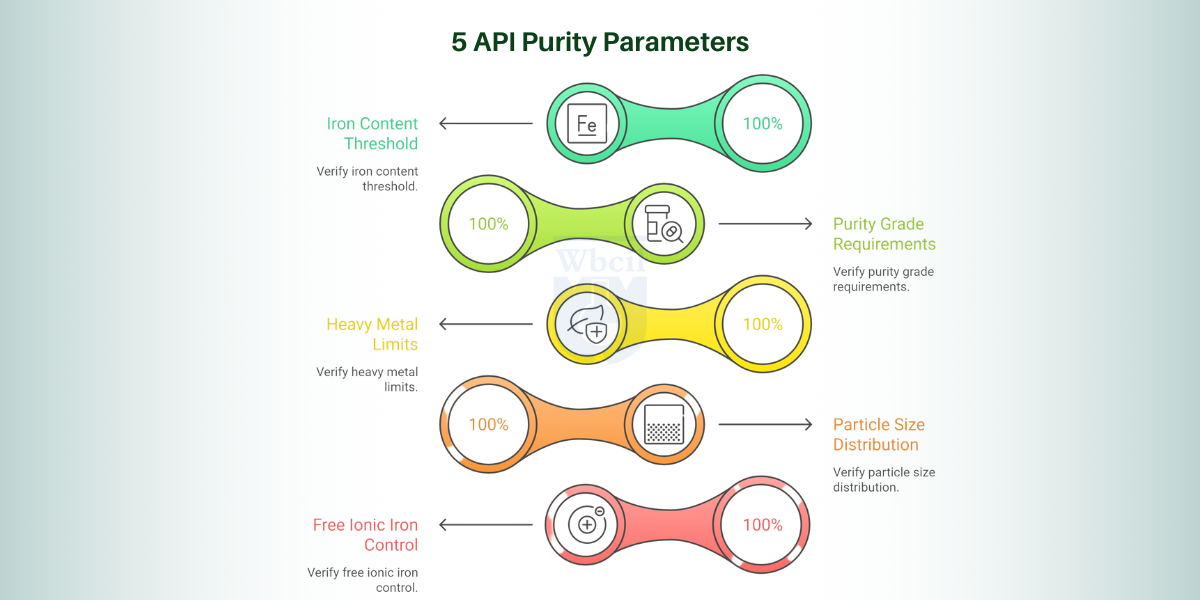
5 Key Purity Parameters Every API Buyer Must Verify
Before you qualify any iron hydroxide polymaltose API batch, five analytical parameters determine whether that material meets pharmaceutical-grade standards for your dosage form.
1. Iron Content Threshold
Iron content must exceed 30% w/w for IPC to perform reliably in haematinic syrup and liquid oral formulations [2]. Below this threshold, your finished product cannot deliver the elemental iron dose claimed on your label. Every batch Certificate of Analysis must confirm this value before your quality team proceeds with qualification.
2. Purity Grade Requirements
Pharmaceutical-grade IPC requires a minimum purity of 98% under rigorous quality-controlled production conditions. Any purity value below this threshold signals process inconsistency and increases the risk of sub-therapeutic batches entering your supply chain.
3. Heavy Metal Limits
Lead, arsenic, and mercury must each fall below 0.002% of total composition per pharmaceutical-grade specifications. Heavy metal contamination in IPC directly elevates patient safety risk, particularly in paediatric syrup and infant drop formulations [3]. Your supplier’s DMF must document validated heavy-metal testing methods and batch-specific results for regulatory submission.
4. Particle Size Distribution
90% of IPC particles must measure below 50 microns to maintain colloidal stability in liquid oral and syrup dosage forms. Particle size directly governs suspension uniformity, which determines dose consistency across every unit your patient receives. Suppliers must provide laser diffraction particle size data as a standard batch release parameter.
5. Free Ionic Iron Control
Free ionic iron, unbound Fe³⁺ not incorporated into the polymaltose complex, is the primary driver of oxidative stress and gastrointestinal toxicity in IPC formulations. Pharmaceutical-grade IPC must maintain free iron at the lowest measurable level to preserve both patient safety and formulation stability. Any supplier unable to provide validated free-iron quantification data should not progress beyond initial qualification.
Also read: 9 Best Applications of Iron(III) Hydroxide Polymaltose Complex (IPC)
Molecular Weight Distribution and Therapeutic Equivalence
Beyond standard purity parameters, molecular weight distribution is the single most consequential quality attribute that separates a therapeutically equivalent IPC from one that merely passes basic iron content tests.
- Lower molecular weight IPC fractions release iron faster, whilst higher MW fractions provide sustained, depot-like iron delivery. This distribution directly determines your finished product’s absorption profile and clinical performance.
- Physicochemical studies confirm that follow-on IPCs exhibit measurable differences in MW distribution compared to the originator product. These differences affect therapeutic equivalence and carry direct implications for your generic submission strategy.
- Proprietary synthesis conditions, including reaction pH, temperature, and ferric salt concentration, directly shape the MW distribution of each IPC batch. Two suppliers using different protocols will consistently deliver non-identical MW profiles.
- Gel permeation chromatography (GPC) or size-exclusion chromatography (SEC) characterisation data is the only reliable method for verifying MW distribution at the batch level. Suppliers unable to provide this data present an unacceptable therapeutic equivalence risk for regulated market submissions [4].
- Your supplier’s Drug Master File must explicitly document the acceptable MW distribution range as a critical quality attribute. Without this specification in the DMF, your regulatory submission has no defensible basis for therapeutic equivalence claims.
In practice, differences in molecular weight distribution often become a key objection during regulatory review. Differences in the safety profiles of iron complexes directly influence both clinical outcomes and regulatory acceptance.
Iron Polymaltose vs. Iron Sucrose: Safety Profile Compared
For generic manufacturers and formulation teams, the safety profile difference between these two iron preparations determines both your patient population scope and your regulatory risk exposure.
| Parameter | Iron Polymaltose Complex (IPC) | Iron Sucrose |
| Iron Form | Non-ionic Fe³⁺, no free ionic iron release | Ionic Fe³⁺ releases free iron into circulation |
| Administration Route | Oral and parenteral formulations are both available | Primarily parenteral administration only |
| GI Tolerability | Lower GI adverse events vs. ferrous salts confirmed | Higher GI disturbance risk than IPC in oral use |
| Hypersensitivity Risk | Rare allergic reactions in 0.01–0.1% of cases [5]. | 0.3-0.4% serious hypersensitivity per FERWON-IDA trial [6]. |
| Food Interaction | No interactions with food or medications known | Must be administered away from food interference |
| Absorption Mechanism | Controlled active absorption, no passive diffusion | Passive diffusion contributes to iron release |
Each parameter here carries direct implications for safety, compliance, and therapeutic consistency.
Sourcing High-Purity Iron Polymaltose API Through WBCIL
When iron hydroxide polymaltose purity benchmarks are this clearly defined, your API supplier’s manufacturing credentials determine whether your finished formulation consistently meets those benchmarks at commercial scale.
West Bengal Chemical Industries Ltd’s legacy spans over 64 years, with IPC produced under WHO-GMP, cGMP, and ISO 9001:2015 certified conditions across facilities in Kolkata and Dahej, Gujarat.
- WBCIL’s IPC delivers iron content above 30% w/w, meeting the pharmaceutical benchmark for haematinic syrup, drops, and liquid oral dosage forms. Every batch release includes validated iron content data, giving your quality team a defensible specification baseline from day one.
- It is suitable for syrup, drops, film-coated tablets, chewable tablets, and liquid oral formulations across paediatric and adult patient segments. This physical form versatility means a single qualified API source covers your entire IPC formulation portfolio.
- WBCIL maintains validated Drug Master Files in CTD format with USFDA DMF submissions already on file. Your regulatory team receives complete, audit-ready documentation for both domestic Indian filings and export market submissions without additional preparation burden.
- WBCIL has consistently supplied IPC to pharmaceutical manufacturers across Japan, Germany, the UK, Australia, and the MENA region. For manufacturers with international market ambitions, this export compliance track record reduces supply chain qualification risk at every regulatory touchpoint.
- Supports bulk procurement of the iron polymaltose API with flexible order quantities, consistent batch-to-batch quality, and full documentation support. Manufacturers who buy iron polymaltose API in bulk from a WHO-GMP-certified source eliminate the quality variability risk that follow-on IPC suppliers often carry.
Also read: Top 5 Frequently Asked Questions About Iron (III) Hydroxide Polymaltose Complex
Final Thoughts
Iron hydroxide polymaltose purity is not a single number on a Certificate of Analysis; it is a multi-parameter quality profile for sourcing and formulation teams. Start by requesting GPC or SEC molecular weight distribution data from every prospective supplier to gather details for the standard purity assay. Ensure your supplier’s DMF explicitly documents MW distribution range, free iron limits, and particle size specifications as critical quality attributes for your regulatory submission.
On the formulation side, match your IPC physical form, powder for liquid oral, solid for tablet compression, to the dosage form requirements before finalising your API specification. WBCIL, a WHO-GMP and cGMP-certified bulk iron polymaltose API supplier with over 64 years of experience, provides your production process with the quality foundation it needs at every batch.
- UNICEF (2024). Frequently asked questions on Anaemia. [online] Unicef.org.
- CPHI (2023). Iron III Hydroxide Polymaltose Complex | CPHI Online. [online]
- Iron, P. (2026). Iron Hydroxide Polymaltose | Drug Information, Uses, Side Effects, Chemistry | PharmaCompass.com. [online] PharmaCompass.com.
- Geisser, P. (2011). Safety and Efficacy of Iron(III)-hydroxide Polymaltose Complex. Arzneimittelforschung, 57(06), pp.439–452.
- Qassim, A., Mol, B.W., Grivell, R.M. and Grzeskowiak, L.E. (2017). Safety and efficacy of intravenous iron polymaltose, iron sucrose and ferric carboxymaltose in pregnancy: A systematic review. Australian and New Zealand Journal of Obstetrics and Gynaecology, 58(1), pp.22–39.
- Auerbach, M., Henry, D., Derman, R.J., Achebe, M.M., Thomsen, L.L. and Glaspy, J. (2019). A prospective, multi‐center, randomized comparison of iron isomaltoside 1000 versus iron sucrose in patients with iron deficiency anaemia; the FERWON‐IDA trial. American Journal of Hematology, 94(9), pp.1007–1014.
Evaluating the API purity of iron polymaltose for generics requires more than just iron content testing. You must verify purity above 98%, control free ionic iron, and confirm particle size and heavy metal limits.
Molecular weight distribution in iron hydroxide polymaltose determines how iron is released and absorbed in the body. Variations in distribution can impact therapeutic equivalence and affect regulatory approval for generic products.
The safety profile of iron polymaltose vs iron sucrose shows that polymaltose has fewer gastrointestinal side effects due to its non-ionic structure. Iron sucrose releases free iron more readily, which can increase the risk of hypersensitivity and adverse reactions.
Sourcing high-purity iron polymaltose for liquid oral dosage requires iron content above 30% w/w and particle size below 50 microns. You must also ensure low free iron levels to maintain stability and reduce gastrointestinal irritation.
The iron hydroxide polymaltose manufacturing process and the impurities it produces depend on reaction conditions, including pH, temperature, and ferric salt concentration. Poor control can lead to inconsistent molecular structure, higher impurities, and reduced product quality.