Identifying Magnesium Bisglycinate: Spotting Low-Grade APIs
Magnesium bisglycinate’s premium positioning in the supplement market has made it a primary target for economically motivated adulteration, a problem that standard quality control protocols are structurally ill-equipped to detect. Recent research confirmed that almost every tested brand failed to contain the chelated magnesium form stated on the label, despite meeting total elemental magnesium requirements.
The adulteration mechanism is straightforward: magnesium oxide blended with glycine passes a standard ICP-OES potency test identically to a fully reacted chelate. The clinical and commercial consequences include reduced bioavailability, GI adverse events, and patient non-compliance, manifest only after the product reaches the market.
In this blog, we examine the chemistry, analytical verification methods, formulation parameters, and procurement criteria required before sourcing a pharmaceutical-grade magnesium bisglycinate API.
Key Takeaways:
- Buffered magnesium bisglycinate passes standard ICP-OES potency testing; only water-soluble extraction or ¹H NMR confirms actual chelation integrity.
- Elemental magnesium above 16% in a claimed pure bisglycinate is a direct indicator of oxide buffering or incomplete chelation reaction.
- Fully reacted bisglycinate absorbs via peptide transport channels, a mechanistically distinct pathway that inorganic oxide blends cannot replicate.
Quick Answer: Identifying magnesium bisglycinate quality requires ¹H NMR or FTIR-ATR chelation confirmation; elemental potency tests alone cannot detect oxide buffering or incomplete chelation in API batches.

What Fully Reacted Magnesium Bisglycinate Really Means
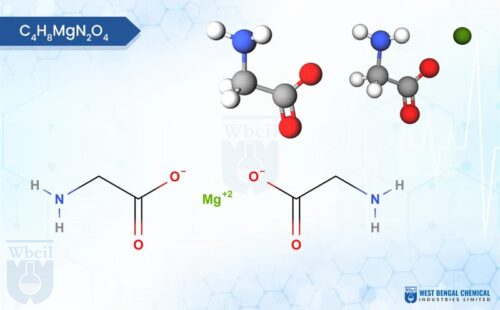
Fully reacted magnesium bisglycinate is a chelate compound with one magnesium ion bonded to two glycine molecules, molecular formula C₄H₈MgN₂O₄, molar mass 172.42 g/mol. Understanding this chemistry is the first step in evaluating the quality of magnesium bisglycinate, because the bond between magnesium and glycine is exactly what low-grade suppliers skip or dilute.
- In fully reacted bisglycinate, each magnesium ion forms a stable heterocyclic ring structure with two glycine molecules via coordinate covalent bonds. The ring shields magnesium from stomach acid, phytates, and competing minerals, none of which is possible in a simple oxide-glycine blend [1].
- Pure magnesium bisglycinate contains approximately 14.1% elemental magnesium by weight; the remaining mass is accounted for by the glycine ligands. Any supplier claiming elemental magnesium above 16% in a pure bisglycinate is almost certainly supplying a buffered or adulterated product.
- The gut recognises chelate as an amino acid complex; it absorbs through peptide and amino acid transport channels in the small intestine. It bypasses the competitive mineral absorption pathways that limit the absorption of inorganic magnesium salts.
- A fully reacted magnesium bisglycinate does not readily absorb atmospheric moisture, unlike inorganic salts. Such non-hygroscopic character directly supports powder flowability, tablet compressibility, and long-term API stability in your manufacturing environment.
- Pharmaceutical-grade magnesium bisglycinate is an off-white to white free-flowing powder with a mild, slightly sweet taste from the glycine component. A gritty texture, chalky appearance, or bitter taste at the raw material stage are physical indicators of oxide contamination or incomplete chelation.
Buffered vs Fully Reacted: Where Most Buyers Get Misled
At the API sourcing stage, quality evaluation becomes a commercial risk decision rather than a routine QC step, because buffered vs fully reacted magnesium bisglycinate appears identical on standard elemental potency tests. A 2024 double-blind, randomised crossover clinical study across 40 healthy volunteers found that magnesium bisglycinate produced no significant plasma increase at any tested time point [2].
Here is a tabular representation to understand the difference between buffered and fully reacted magnesium bisglycinate:
| Parameter | Buffered | Fully Reacted | B2B Risk |
| Composition | Magnesium oxide blended with glycine, no covalent bond formed | One magnesium ion is covalently bonded to two glycine molecules | Standard CoA cannot distinguish the two without spectroscopic testing |
| Elemental magnesium content | Artificially elevated at 18-20% due to high oxide magnesium density | Approximately 14.1% glycine ligands account for the remaining mass | Any supplier claiming above 16% in a pure bisglycinate is supplying a blend |
| Water solubility | Partially soluble, oxide fraction produces visible sedimentation in liquids | Fully water-soluble, chelate disperses completely in aqueous matrices | Sedimentation in liquid formats is an observable quality failure requiring no lab |
| GI tolerability | Oxide fraction causes an osmotic laxative effect and reduces patient adherence | Absorbs via peptide transport channels with no osmotic interference | Consumer GI complaints in finished products trace directly to a buffered API source |
| Detection method | Passes standard ICP-OES elemental testing; adulteration is invisible at the potency level. | Requires water-soluble magnesium extraction or ¹H NMR to confirm chelation | Without chelation verification in your CoA requirements, a buffered product passes incoming QC |
For procurement teams, this difference directly affects both product performance and long-term brand trust.
Also read: 10 Top Benefits of Magnesium Bisglycinate
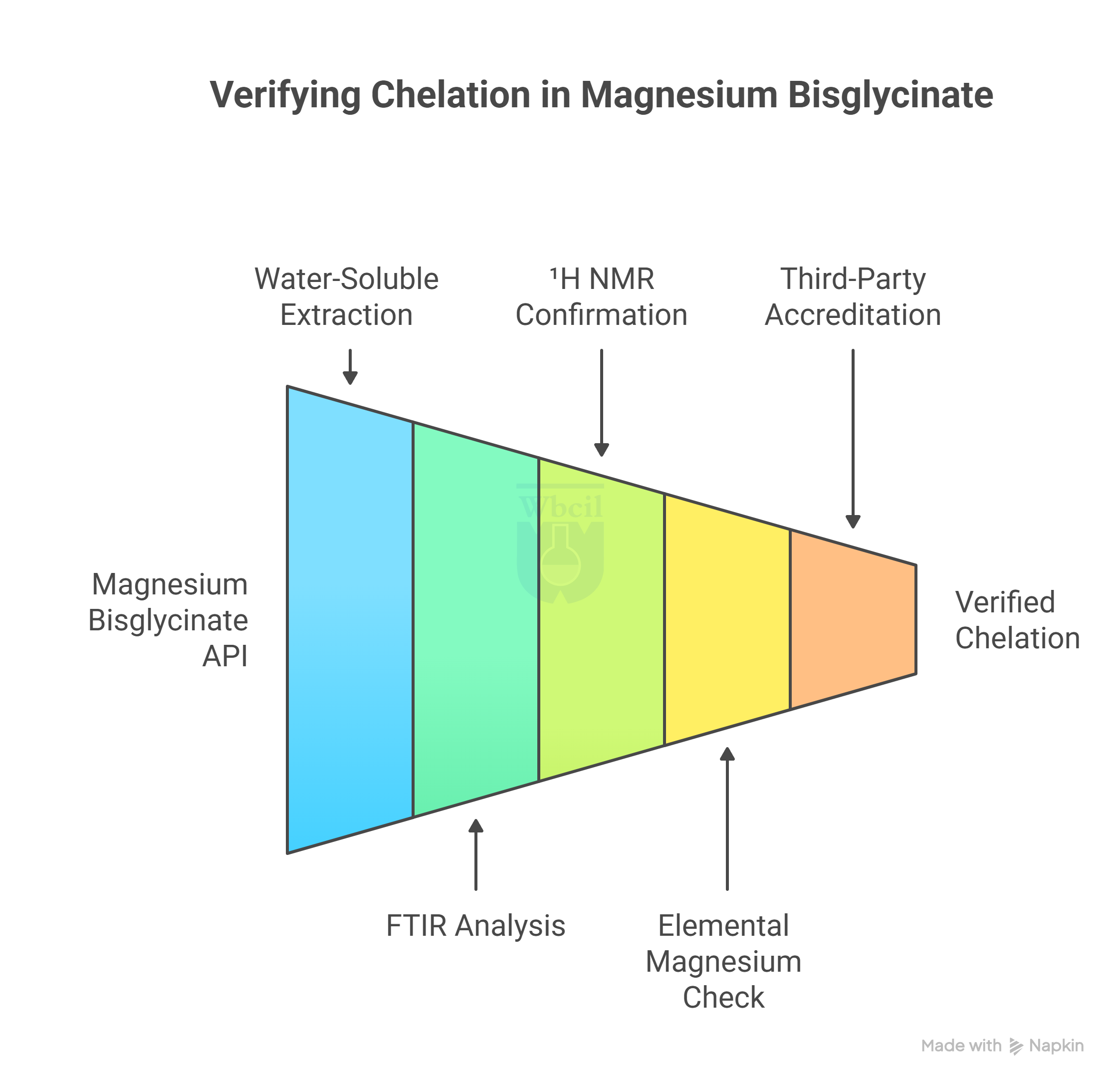
How to Verify Chelation in a Magnesium Bisglycinate API
Verifying chelation in a magnesium bisglycinate API requires a sequential testing approach. Moreover, no single method confirms bond integrity, purity, and elemental accuracy together.
Here’s a step-by-step process to verify the chelation in magnesium bisglycinate API:
Step 1: Run a Water-Soluble Magnesium Extraction First
Standard ICP-OES testing dissolves the entire sample, and both chelated and unchelated magnesium appear as the same value. A water extraction step applied before ICP-OES separates soluble chelated magnesium from insoluble oxide fractions. This additional step exposes differences that standard potency testing cannot detect.
Step 2: Use FTIR to Check the Bond Signature
FTIR spectroscopy reads the molecular fingerprint of a solid API sample without any preparation or dissolution. A genuine magnesium bisglycinate chelate produces specific infrared absorption peaks from the carboxylate and amino groups bonded to magnesium. A dry oxide-glycine blend produces a different spectrum entirely; the chelation bond peaks are simply absent [3].
Step 3: Confirm Bond Integrity With ¹H NMR
¹H NMR is the only test that confirms the actual covalent bond between magnesium and glycine at the molecular level. FTIR tells you a bond exists, and NMR tells you how much of your sample is actually chelated versus unreacted.
Step 4: Cross-Check the Elemental Magnesium Percentage
Pure magnesium bisglycinate contains approximately 14.1% elemental magnesium by weight; this is a fixed chemical property of the chelate. If a supplier reports elemental magnesium above 16% in a claimed pure bisglycinate, the batch contains oxide buffering or incomplete reaction. Pairing this number with the water-soluble magnesium fraction provides two-point verification that captures what either test alone misses.
Step 5: Require Third-Party ISO/IEC 17025 Accredited Results
A supplier’s CoA reflects only the parameters they chose to test, not necessarily the full quality profile of the batch. Third-party verification from an ISO/IEC 17025 accredited laboratory confirms identity, chelation integrity, and potency under a recognised quality standard.
Also read: Overcoming Regulatory Hurdles: WBCIL’s Approach to GMP and Safety Compliance.
Solubility and Stability in Liquid Dosage Forms
Magnesium bisglycinate’s suitability for liquid formats extends beyond solubility; it also offers distinct stability and sensory properties.
- Unlike inorganic salts, fully reacted bisglycinate does not absorb moisture from ambient air. It prevents clumping, preserves powder flowability, and reduces the risk of degradation in your production line.
- Pharmaceutical-grade magnesium bisglycinate is designed for excellent flowability and compressibility in tablet manufacturing. Poor flowability causes content uniformity failures, a problem bisglycinate’s physical profile avoids by design.
- Magnesium bisglycinate remains chemically stable in formulations containing multiple actives and excipients, across varying pH values.
- In vitro studies confirm bisglycinate releases magnesium ions faster than oxide under fasted stomach conditions. Patients often take supplements without food; your label claims must account for this.
- Magnesium bisglycinate has no metallic taste and produces no bitter off-notes. This makes it suitable for syrups, oral solutions, and semi-liquid gels for elderly patients.
Identifying the quality of magnesium bisglycinate at the procurement stage starts with knowing exactly which documentation to demand.
What to Demand From Your API Supplier
Sourcing pharmaceutical-grade magnesium bisglycinate requires supplier documentation that covers chelation integrity, in vitro dissolution data, and regulatory readiness.
- Demand ¹H NMR or FTIR-ATR chelation confirmation alongside in vitro water-soluble magnesium extraction data per batch.
- Your supplier should provide in vivo bioavailability references aligned with your target dosage format (capsule, syrup, or gummy).
- Reject any batch reporting elemental magnesium above 16% in a claimed pure pharmaceutical-grade magnesium bisglycinate without written justification.
- West Bengal Chemical Industries Limited supplies market-specific dossiers, white papers, and, upon request, access to the manufacturer’s sample of the magnesium bisglycinate API.
Final Thoughts
Assessing magnesium bisglycinate quality cannot rely only on supplier declarations or standard potency reports. You need clear evidence of chelation integrity through spectroscopic testing and verified batch-level documentation before committing to procurement. As regulatory expectations continue to tighten across CDSCO, USFDA, and FSSAI frameworks, your sourcing decisions must prioritise chemical identity, stability data, and compliance readiness from the outset.
Working with a manufacturing partner that treats chelation verification and batch transparency as standard practice significantly reduces both quality and regulatory risk. WBCIL’s pharmaceutical-grade magnesium bisglycinate is built around this approach, supported by certified documentation, regulatory dossiers, and consistent batch verification.
- Case, D. R., Zubieta, J., Gonzalez, R. and Doyle, R. P. (2021). Synthesis and Chemical and Biological Evaluation of a Glycine Tripeptide Chelate of Magnesium. [online] National Library of Medicine.
- Pajuelo, D., Meissner, J.M., Negra, T., Connolly, A. and Mullor, J.L. (2024). Comparative Clinical Study on Magnesium Absorption and Side Effects After Oral Intake of Microencapsulated Magnesium (MAGSHAPETM Microcapsules) Versus Other Magnesium Sources. Nutrients, 16(24), p.4367.
- Miller, M.E., McKinnon, L.P. and Walker, E.B. (2015). Quantitative measurement of metal chelation by Fourier transform infrared spectroscopy. Analytical Chemistry Research, 6, pp.32–35.
Chelation verification requires ¹H NMR or FTIR-ATR spectroscopic testing, not standard ICP-OES potency analysis. Water-soluble magnesium extraction, combined with ICP-OES, provides a second confirmation step that separates chelated from unchelated fractions.
¹H NMR is the gold standard for purity testing; it confirms bond integrity and quantifies unreacted oxide fractions simultaneously. Third-party ISO/IEC 17025-accredited results provide an independent verification layer that internal CoA documentation cannot replace.
Fully reacted magnesium bisglycinate is water-soluble and pH-stable across the 4.0-6.5 range typical of oral syrups and solutions. A buffered or partially reacted batch introduces insoluble oxide particles that cause visible sedimentation, which is a direct quality failure in liquid formats.
Bulk sourcing decisions should prioritise batch-level chelation confirmation, elemental magnesium percentage within the 14.1% range, and ICH-compliant stability data. CGMP and ISO certification with third-party audit records are non-negotiable procurement criteria for pharmaceutical and nutraceutical applications.
Elemental magnesium above 16% in a claimed pure bisglycinate is the primary label-level indicator of oxide blending. Water-soluble magnesium extraction via ICP-OES or ¹H NMR at a certified laboratory confirms molecular-level adulteration.