Clean Label Iron Supplements: The Liposomal Iron Advantage
Iron deficiency affects nearly a third of the global population, and the supplement industry’s standard answer, ferrous sulfate, has a compliance problem that formulators can no longer ignore. If you have ever watched a well-designed iron product fail in the market because patients stopped taking it, you already understand the core issue. The gap between elemental iron on a label and iron that actually reaches the bloodstream is where most conventional formulations break down. The liposomal iron advantage addresses that gap at the structural level by using phospholipid encapsulation to alter how iron behaves in the body.
In this blog, we examine the clinical evidence, formulation science, and sourcing considerations that B2B buyers need before committing to a high bioavailability iron API for their next product.
Key Takeaways:
- Ferrous salts absorb at 10-15% of the ingested dose; WBCIL’s Lipoedge TM liposomal iron reaches cells via endocytosis, bypassing the DMT1 absorption ceiling entirely.
- Clinical data from India and internationally confirm that liposomal iron delivers faster haemoglobin recovery at a fraction of the conventional elemental dose.
- Format compatibility, sachets, gummies, RTD beverages, is where liposomal iron creates product differentiation that ferrous salts structurally cannot.
Quick Answer: Liposomal iron absorbs via endocytosis, bypasses DMT1 limits, delivers superior haemoglobin outcomes at lower doses with far better tolerability.

Why Traditional Iron Salts Fail Formulators
Iron deficiency is one of the major causes of anaemia, affecting roughly 27% of individuals globally, according to the WHO [1]. However, ferrous sulfate, which is a prominent medication, has serious compliance problems. For formulators in the pharmaceutical and nutraceutical sectors, it creates product-level liability.
Here are some of the factors that contribute to the lack of efficacy of traditional iron salts:
1. Lower Absorption Rate
Ferrous salts are absorbed primarily in the duodenum via the DMT1 transporter protein. Such a carrier-mediated pathway is narrow, saturable, and highly sensitive to competing dietary factors. A clinical review confirms that the absorption rate of ferrous salts is 10-15%, with no difference between ferrous sulfate, fumarate, or gluconate preparations [2].
- GI Side Effects
A systematic review and meta-analysis found that ferrous sulfate supplementation increased the risk of GI side effects, with an odds ratio of 2.32 compared with placebo [3]. Nausea, constipation, epigastric pain, and metallic taste are not rare; adherence to oral iron therapy hovers at only 40-60%. A product that patients stop taking cannot correct iron deficiency, regardless of its elemental iron content.
3. Enteric-Coated Formulations
Formulators prefer enteric-coated preparations to reduce overall GI irritation. The bioavailability of enteric-coated ferrous sulfate is estimated at just 30% of the standard oral preparation, because iron released beyond the duodenum remains largely unabsorbed. The attempt to fix one problem creates another, and a B2B buyer ends up with an API that is neither well-tolerated nor well-absorbed.
4. Formulation Problem
Phytates in whole grains, polyphenols in tea and coffee, and calcium from dairy all bind free iron in the gut, preventing its absorption. India’s predominantly plant-based diet means these inhibitors are consistently present. A formulation built on ferrous salts must account for this interference, and there is no practical API-level solution within the conventional salt framework.
5. Hepcidin Blocks Absorption
The liver hormone hepcidin binds to ferroportin, the only known cellular iron exporter, and causes its degradation, reducing intestinal iron absorption [4]. In patients with chronic inflammation, kidney disease, or infection, hepcidin levels stay elevated. Ferrous salts have no mechanism to bypass this blockade, making them especially unreliable in the patient populations that need iron supplementation most.
Before you source an iron API, you need to understand what liposomal iron delivery actually does at the cellular level.
How Liposomal Iron Delivery Actually Works
Liposomes are nanoscale spheres formed from phospholipid bilayers, the same material that makes up human cell membranes. When iron, typically as ferric pyrophosphate, is encapsulated within this bilayer, it travels through the gastrointestinal tract in a chemically protected state.
The protection removes the primary reasons ferrous salts fail: no direct contact with the gut lining, no reaction with dietary inhibitors, and no dependency on them. For pharma and nutraceutical manufacturers, this is an API with a mechanistically distinct and clinically superior absorption pathway.
The following sections provide a comprehensive overview of how liposomal technology provides an advantage at the structural levels:
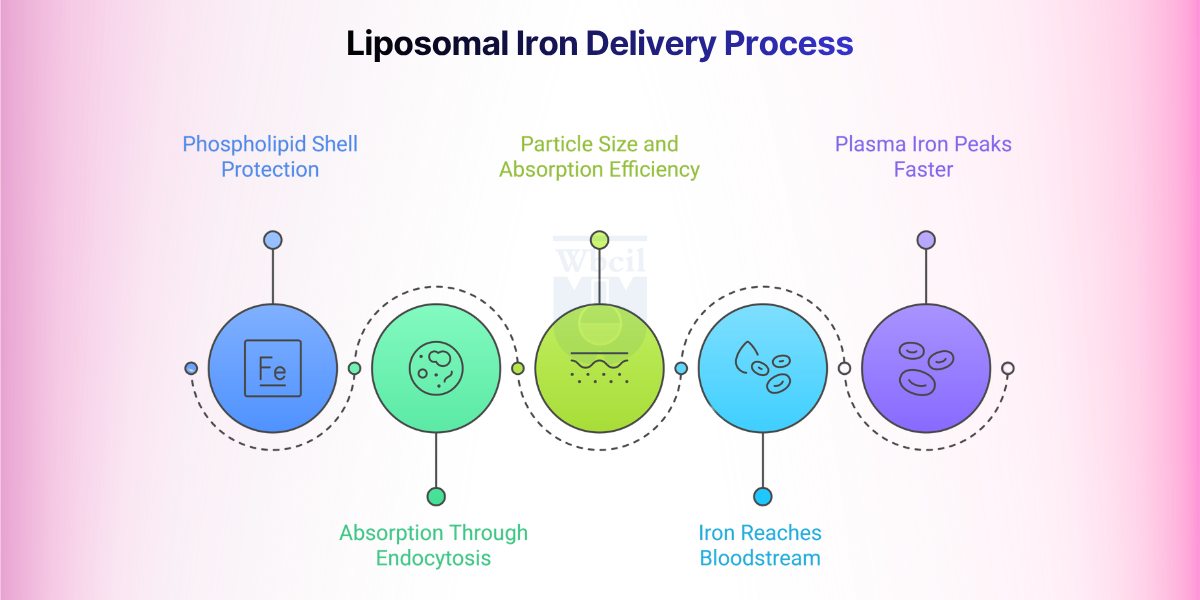
1. The Phospholipid Shell Protection
Once ingested, the phospholipid bilayer of liposomal iron resists breakdown by stomach acid, bile salts, and pancreatic lipases. The liposome’s outer membrane acts as a chemical barrier, keeping the iron payload intact until it reaches the small intestine. A 2019 study on liposomal delivery confirmed that the lipid bilayer prevents iron oxidation and enzymatic degradation before absorption occurs [5].
2. Absorption Occurs Through Endocytosis
Unlike ferrous salts that depend on DMT1-mediated transport, liposomal iron enters intestinal cells through endocytosis. Research on hepcidin, DMT1, and ferroportin regulation published in PMC confirms that liposomal encapsulation circumvents the standard iron absorption gateways entirely [6].
3. Particle Size and Absorption Efficiency
Liposomes in the 30-100 nm range achieve optimal uptake through intestinal M-cells in Peyer’s patches. Particles outside this size range absorb inconsistently. Accelerated stability studies confirm encapsulation efficiency above 85%. Zeta potential remains approximately −32 mV at pH 7.0.
4. Iron Reaches the Bloodstream Without Oxidative Damage
Free iron from ferrous salts generates reactive oxygen species (ROS) on contact with the intestinal mucosa. This oxidative stress is a direct cause of GI side effects. Liposomal encapsulation prevents this contact entirely. A comparative tolerability study confirmed that microencapsulated iron pyrophosphate formulations reversed iron deficiency within 14 days in animal models with no adverse GI effects [7]. The iron payload is delivered directly to the lymphatic system, bypassing first-pass hepatic exposure and reducing the risk of systemic iron overload.
5. Plasma Iron Peaks Faster at Lower Doses
Liposomal iron reaches maximum plasma concentration within two hours of oral administration. Clinical data on liposomal mineral absorption showed a superior pharmacokinetic profile for liposomal iron compared with standard formulations at equivalent or lower elemental iron doses [8]. For a formulator, this means a lower-dose product can achieve the same or better therapeutic outcome, a commercial and regulatory advantage when positioning a clean-label supplement.
Also read: Decoding the Science: Key Characterisation Parameters of Liposomal Iron.
Liposomal Iron vs Iron Salts: Bioavailability
Every API sourcing decision carries downstream product risk. When your formulation depends on ferrous sulfate, that risk sits in a documented gap between the elemental iron dose you put into a capsule and what a patient’s body actually uses.
Ferrous salts are inexpensive and clinically established, but their absorption ceiling and dose-driven compliance issues make the product commercially underperform before it underperforms clinically.
| Parameter | Ferrous Salts | WBCIL’s Lipoedge TM Liposomal Iron |
| Effective daily dose | High elemental iron dose required to compensate for poor gut absorption. | Clinically effective at a fraction of the conventional dose, with comparable or superior haemoglobin outcomes. |
| Haemoglobin recovery | Consistent across populations, but response plateaus due to absorption constraints after each dose. | Faster haemoglobin recovery at a lower elemental dose; clinical advantage is most pronounced in women and children. |
| Ferritin repletion | Strong and well-documented across adult and paediatric groups; a reliable marker for iron store recovery. | Haemoglobin and serum iron recovery are reliable; ferritin repletion is more variable, particularly in children. |
| Patient adherence | A large share of patients reduce the dose or withdraw entirely due to tolerability issues, which directly undermines treatment outcomes. | Significantly higher adherence in clinical settings; superior oral tolerability and lower incidence of constipation confirmed in an Indian paediatric RCT. |
| Formulation compatibility | Metallic taste, pH sensitivity, and colour instability limit ferrous salts to tablet and capsule formulations in most manufacturing contexts. | No metallic taste; stable across sachets, gummies, RTD beverages, and fortified foods without sensory compromise. |
Understanding the liposomal iron advantage means knowing exactly where it fits within your nutraceutical product architecture.
Liposomal Iron in Nutraceutical Formulations
Lipoedge TM Liposomal iron’s format neutrality, no metallic taste, no colour interference, stable across pH ranges, is what separates it from every conventional iron API when it comes to nutraceutical product development.
- Liposomal iron is available as liquid liposomes for sprays and as powdered pro-liposomes for gummies and sachets. Ferrous salts cannot be used in these formats due to their metallic taste and colour reactivity in food matrices.
- Three out of four Indian women have low dietary iron intake, per market research. Prenatal sachets, adolescent gummies, and postpartum blends all require an iron API without sensory compromise.
- Liposomes in aqueous form are vulnerable to leakage, oxidation, and aggregation under heat and mechanical stress. Stability data under your specific processing conditions is a primary quality parameter, not optional documentation.
- Sports nutrition needs iron replenishment at a lower dose without disrupting training or digestion. Paediatric formats need to be palatable and free of a metallic taste, as this is the main reason parents reduce or skip doses.
- Liposomal iron sourced from a WHO-GMP certified manufacturer meets FSSAI clean-label requirements technically. Your API supplier’s regulatory dossier support is as commercially important as the ingredient specification itself.
- Liposomal iron functions as a targeted nutrient delivery system, directing iron payloads to absorption sites with minimal systemic loss.
Your search for the liposomal iron advantage ends with a manufacturer whose science and supply chain match your brief.
Sourcing Liposomal Iron API from WBCIL: A B2B Guide
West Bengal Chemical Industries Limited supplies a pharma-grade, non-constipating iron API built on patented encapsulated iron technology — backed by 64 years of manufacturing depth.
- WBCIL’s Lipoedge TM liposomal iron portfolio covers ferric pyrophosphate, ferrous bisglycinate, ferrous ascorbate, and ferrous sulphate in encapsulated form. Each salt targets a distinct clinical or formulation brief, giving your product team precise API selection flexibility.
- WBCIL’s microencapsulated iron pyrophosphate achieves encapsulation efficiency above 85%, confirmed by HPLC across all commercial batches. Vesicles are engineered at 200-300 nm with a PDI of ≤0.25 and zeta potential above ±30 mV for colloidal stability.
- The API is available as bulk powder or a liquid colloidal suspension, compatible with capsules, sachets, gummies, syrups, and sublingual tablets. If you buy liposomal iron API in bulk, WBCIL supplies it in food-safe HDPE drums with a confirmed shelf life of 3 years.
- Every batch carries full CoA, ICH Q1A/Q1B stability data, and dossiers prepared for USFDA, EMA, CDSCO, and FSSAI submissions. WBCIL holds 17 active patents, including a patented solvent-free green liposomal manufacturing process.
- WBCIL operates under WHO-GMP, cGMP, ISO, HACCP, and FSSAI certification across its Kolkata and Dahej manufacturing facilities. Over 4,000 clients across 30+ countries source iron from West Bengal Chemical Industries Limited.
Final Thoughts
The evidence across this blog points to one clear conclusion: the choice of iron API is a product strategy decision, not just a chemistry decision. If your target segment includes women’s health, maternal nutrition, paediatric formats, or functional foods, the liposomal iron advantage is not incremental. Before you finalise your next iron formulation, request encapsulation efficiency data, ICH-compliant stability reports, and multi-market regulatory dossiers from your API supplier. These documents determine your product’s commercial shelf life.
Sourcing a high bioavailability iron API from a WHO-GMP and cGMP certified manufacturer with documented batch-to-batch consistency removes the single largest post-launch risk in iron supplement development. It gives buyers a validated pathway to faster product development, regulatory approval, and market success.
- Owaidah, T., Al-Numair, N., Al-Suliman, A., Zolaly, M., Hasanato, R., Al Zahrani, F., Albalawi, M., Bashawri, L., Siddiqui, K., Alalaf, F., Almomen, A. and Sajid, M.R. (2020). Iron Deficiency and Iron Deficiency Anemia Are Common Epidemiological Conditions in Saudi Arabia: Report of the National Epidemiological Survey. Anemia, 2020, pp.1–8.
- Fei, C. (2015). Iron Deficiency Anemia: A Guide to Oral Iron Supplements – Clinical Correlations. [online] Clinical Correlations.
- Tolkien, Z., Stecher, L., Mander, A.P., Pereira, D.I.A. and Powell, J.J. (2015). Ferrous Sulfate Supplementation Causes Significant Gastrointestinal Side-Effects in Adults: A Systematic Review and Meta-Analysis. PLOS ONE, [online] 10(2), p.e0117383.
- Camaschella, C., Nai, A. and Silvestri, L. (2020). Iron metabolism and iron disorders revisited in the hepcidin era. Haematologica, 105(2), p.haematol.2019.232124.
- Maladkar, M., Sankar, S. & Yadav, A. (2020). A Novel Approach for Iron Deficiency Anaemia with Liposomal Iron: Concept to Clinic. Journal of Biosciences and Medicines.
- Przybyszewska, J. and Żekanowska, E. (2014). The role of hepcidin, ferroportin, HCP1, and DMT1 protein in iron absorption in the human digestive tract. Gastroenterology Review, 4, pp.208–213.
- Mariné-Casadó, R., Yaiza Tobajas, Antolín, A., Negra, T., Connolly, A., Alcaide-Hidalgo, J.M. and Antoni Caimari (2025). Comparative study of the effects of different iron sources on bioavailability and gastrointestinal tolerability in iron-deficient rats. Scientific Reports, [online] 15(1).
- Tinsley, G.M., Harty, P.S., Stratton, M.T., Siedler, M.R. and Rodriguez, C. (2022). Liposomal Mineral Absorption: A Randomized Crossover Trial. Nutrients, 14(16), p.3321.
Liposomal iron encapsulates ferric pyrophosphate within a phospholipid bilayer, delivering iron via endocytosis rather than standard gut absorption. This makes it more bioavailable and significantly better tolerated than conventional iron salts.
Liposomal iron produces comparable or superior haemoglobin outcomes at a lower elemental dose with fewer GI side effects. For sensitive populations, pregnant women, children, and IBD patients, it is often considered a better-tolerated option in sensitive populations.
Yes, liposomal iron has no metallic taste and no colour reactivity in food matrices. This makes it compatible with gummies, sachets, syrups, and RTD beverages where ferrous salts cannot be used.
Every batch is tested for encapsulation efficiency, particle size, zeta potential, and stability under WHO-GMP and cGMP certified conditions. Full CoA and ICH-compliant stability data accompany each bulk shipment.
WBCIL prepares dossiers aligned with FDA, EMA, CDSCO, and FSSAI requirements, backed by 17 active patents. B2B buyers receive complete documentation to launch across India and international markets without reformulation.
Yes, liposomal iron is designed to reduce gastrointestinal irritation by preventing direct contact between iron and the gut lining. This makes it suitable for patients who experience intolerance to conventional iron salts.