-
Product Name:
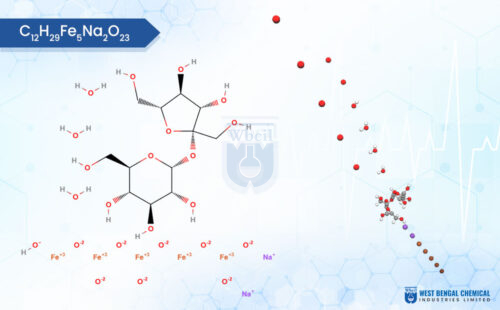
Liposomal Iron
-
Molecular Weight:
745.21 g/mol
-
CAS No.:
10058-44-3
-
HSN Code:
2835
-
Shelf Life:
3 years
-
Salts Available
- Liposomal Ferric Pyrophosphate
- Liposomal Ferrous Bisglycinate
- Liposomal Ferrous Ascorbate
- Liposomal Ferrous Sulphate
- USP
- Insights
- MSDS
- White Paper
USP of Liposomal Iron
- Mimic biological structures
- Highest absorption due to lipid bilayer leading to highest delivery to cells.
- Negligible GI side effect as not absorbs in stomach
- Avoid iron overload in liver by bypassing it
- Replenish iron stores quicker than traditional supplements
- High encapsulation efficiency exceeding 85%.
- Liposomal iron is highly stable (little leakage rate).
Insights Liposomal Iron
- Appearance: Off white dry powder
- Solubility: Water-dispersible (liposomal form)
- Stability: Highly stable under GMP storage conditions
- Formulation Type: Liposomal Suspension / Powder/ Capsule/ Sachet/ Sublingual Tablet/ Gummies/ Syrup
- Particle Size: 100-220 nm
- pH: 6.0-7.5
- Storage Temp: 20°C
- Storage Conditions: Store in a cool, dry place, away from light
- Odor: Mild, slightly earthy odor
- Packaging: Food-safe HDPE drums with inner poly liner
MSDS of Liposomal Iron
Download MSDS PDF- INHALATION: Move exposed person to fresh air. If not breathing, seek immediate medical attention. If breathing is irregular or if respiratory arrest occurs, provide artificial respiration or oxygen by trained personnel and seek medical attention.
- INGESTION: Do not induce vomiting unless directed to do so by medical personnel. Never give anything by mouth to an unconscious person. Seek medical attention.
- SKIN CONTACT: Remove contaminated clothing and shoes and immediately flush skin with plenty of water for at least 15 minutes. Wash clothing before reuse. Clean shoes thoroughly before reuse. If irritation persists, seek medical attention.
- EYE CONTACT: Check for and remove any contact lenses. Immediately flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids.
- HAZARDOUS COMBUSTION PRODUCTS: Decomposition products may include oxides of carbon and iron.
- FIRE: Not considered a fire hazard.
- EXPLOSION: Not considered an explosion hazard.
- FIRE EXTINGUISHING MEDIA: Use fire-extinguishing media appropriate to the surrounding fire.
- SPECIAL INFORMATION: In the event of a fire, wear full protective clothing and NIOSH approved self-contained breathing apparatus with full-face piece operated in the pressure demand or other positive pressure mode.
- ENGINEERING CONTROLS: Use process enclosures, local exhaust ventilation, or other engineering controls to keep airborne levels below recommended exposure limits. If user operations generate dust, fume or mist, use ventilation to keep exposure to airborne Contaminants below the exposure limit.
- PERSONAL PROTECTION: Safety glasses, Lab Coat, Dust respirator, be sure to use an approved / certified respirator or equivalent, Gloves
- PERSONAL PROTECTION IN CASE OF A LARGE SPILL: Splash goggles. Full suit. Dust respirator. Boots. Gloves. A self-contained breathing apparatus should be used to avoid inhalation of the product. Suggested protective clothing might not be sufficient; consult a specialist BEFORE handling this product.
- EXPOSURE LIMITS: Consult local authorities for acceptable exposure limits.
- Appearance Form: Powder
- Color: White yellowish
- Odour: Characteristic
- Solubility: NA
Description of Liposomal Iron
Liposomal iron is a next-generation iron delivery system where elemental iron, as ferric pyrophosphate, ferrous bisglycinate, or ferrous ascorbate, is encapsulated within nanoscale phospholipid vesicles. Structurally identical to human cell membranes, this bilayer architecture makes liposomal iron inherently biocompatible, absorption-ready, and chemically stable across gastric transit. Sourced by reliable liposomal iron ingredient suppliers and wholesale manufacturers globally, liposomal iron is the preferred raw ingredient for companies producing premium iron supplement products.
Available Forms:
Supplied in powder or liquid colloidal suspension, suitable for hard capsules, tablets, softgels, oral drops, and sachet blends. Encapsulation Efficiency % (EE%) exceeds 85%, as confirmed by HPLC quantification across all commercial batches.
Application of Liposomal Iron
- Iron Supplementation: Used in oral liposomal iron supplements to treat iron deficiency anemia, offering improved gastrointestinal tolerance and better absorption compared to traditional iron supplements.
- Chronic Conditions: Beneficial for patients with chronic kidney disease, inflammatory bowel disease, or those undergoing bariatric surgery, as it helps maintain adequate iron levels with fewer side effects.
- Pregnancy: Recommended for pregnant women to prevent and treat iron deficiency anemia, providing a gentler alternative to traditional iron supplements.
- Fortification: Added to food and beverage products to enhance their iron content, especially in products targeted at populations prone to iron deficiency, such as children and pregnant women.
- Athlete Supplements: Included in supplements for athletes to support optimal iron levels, which are crucial for maintaining endurance and overall performance.
- Child Supplements: Used in pediatric formulations to treat iron deficiency anemia in children, providing a more palatable and less irritating option for young patients.
- Animal Health: Incorporated into veterinary supplements to treat and prevent iron deficiency anemia in pets and livestock, improving overall health and growth.
- Elderly Care: Employed in supplements for the elderly to address iron deficiency, offering a more tolerable option for those with sensitive digestive systems.
- IBD Management: Beneficial for patients with inflammatory bowel disease (IBD) as it provides iron supplementation with minimal gastrointestinal side effects, making it suitable for long-term use.
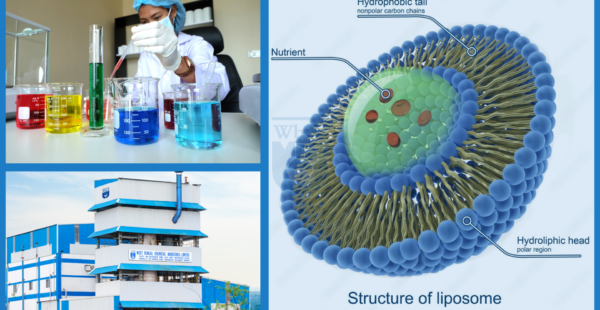
Liposomal Technology & Delivery Science
Liposomes encapsulate active ingredients within a phospholipid bilayer, mirroring natural cell membranes. High-purity lecithin, with phosphatidylcholine and phosphatidylethanolamine content exceeding 90%, governs vesicle performance and stability. The architecture enables direct endocytic cellular uptake, surpassing passive diffusion of traditional iron salts.
Phospholipid Architecture:
90% pure phosphatidylcholine and phosphatidylethanolamine, non-GMO sunflower lecithin, uniform vesicles, consistent EE%, and GMP-grade batch reproducibility.
Vesicle Engineering:
200-300 nm vesicles, PDI ≤0.25, zeta potential ≥ ±30 mV, engineered for consistent bioavailability and stable OEM-grade bulk supply.
Analytical Compliance:
EE% by HPLC, ICH Q1A/Q1B stability, DLS, FTIR, DSC, CoA, full regulatory documentation for FDA, EMA, CDSCO, and FSSAI submissions.
Liposomal Iron Vs Traditional Iron
A side-by-side comparison highlighting how liposomal iron delivery offers improved absorption, enhanced tolerance, and better formulation stability when compared with conventional iron salts.
| Parameter | Liposomal Iron | Traditional Iron Salts |
|---|---|---|
| Absorption Mechanism | Endocytosis, direct cellular uptake | Passive diffusion via the gut wall |
| Bioavailability | High, lipid-mediated transport | Low to moderate |
| GI Tolerability | Minimal, iron shielded from gut lining | Frequent GI discomfort reported |
| Metallic Aftertaste | None, iron fully encapsulated | Common consumer complaint |
| Oxidative Stability | High, protected by a phospholipid shell | Susceptible to degradation |
OEM, Private Label & Bulk Ingredient Supply
WBCIL is a WHO-GMP and cGMP certified liposomal iron manufacturer supplying nutraceutical ingredient bulk, OEM dietary supplements, and private label solutions to supplement companies, contract formulators, and wholesale ingredient buyers. Trusted by reputable brands and supplement companies as a reliable liposomal iron bulk supplier, from raw ingredient sourcing to finished product manufacturing
Contract Manufacturing
End-to-end production with full batch CoA, stability data, and regulatory dossiers
Private Labelling in 30+ Countries
Flexible MOQ available, custom packaging and label design, backed by 64 years of formulation expertise
Bulk Ingredient Supply
WHO-GMP-certified liposomal iron powder in HDPE drums.
Sourcing Liposomal Iron from WBCIL
- Manufacturing Legacy: 64+ years of pharmaceutical API and liposomal supplement manufacturing experience across India’s most regulated facilities.
- Patented Science: 17 Active Patents in liposomal and API technology, including a patented solvent-free green liposomal manufacturing process.
- Global Reach: Trusted by 4,000+ clients, including global nutraceutical OEM manufacturers, formulators, and reputable supplement companies.
- Award Recognised: National IP Award 2024 winner, India’s Top MSME for Patent Filing and Commercialisation by the Ministry of Commerce & Industry.
- Certified Compliance: WHO-GMP, cGMP, ISO, HACCP, and FSSAI certified, with a USFDA guideline-maintained production process.
Liposomal iron encapsulates elemental iron within nanoscale phospholipid vesicles, delivering it via endocytosis rather than passive gut diffusion, resulting in higher bioavailability and significantly reduced GI side effects versus traditional iron salts.
WBCIL brings 64+ years of pharmaceutical manufacturing experience, 14 active patents, including a solvent-free, green liposomal process, and certifications under WHO-GMP, cGMP, ISO, and HACCP. Trusted by 4,000+ clients across 30+ countries, WBCIL delivers full regulatory dossiers and batch CoAs, making it a reliable partner for nutraceutical OEMs and private-label buyers globally.
Key quality parameters include encapsulation efficiency exceeding 85%, particle size between 20 and 300 nm, PDI ≤ 0.25, and zeta potential ≥ ±30 mV, validated by HPLC, DLS, and ICH Q1A/Q1B stability studies, with full CoA documentation.
Liposomal iron is significantly better tolerated than conventional iron supplements. The phospholipid bilayer shields the gut lining from direct contact with iron, minimising constipation, nausea, and a metallic aftertaste commonly associated with ferrous sulphate and ferric salts.
WBCIL supplies liposomal iron as a bulk powder or a liquid colloidal suspension, compatible with hard capsules, tablets, softgels, oral drops, sachet blends, and other formats. OEM contract manufacturing, private-label with custom packaging, and raw ingredient supply are available with flexible MOQs.
Every batch is manufactured under WHO-GMP and cGMP certified conditions, supported by ICH Q1A/Q1B stability data, HPLC-verified encapsulation efficiency, and complete CoA documentation. Regulatory dossiers are prepared to meet FDA, EMA, CDSCO, and FSSAI submission standards, enabling supplement companies to launch confidently across multiple international markets without reformulation delays.