WBCIL’s Adherence to USFDA Guidelines in API Manufacturing
Regulatory compliance is one of those things that sounds straightforward on paper but is genuinely difficult to get right in a factory. It takes consistent discipline — not just during an audit, but on every production run, in every batch record, and across every gram of material that leaves your facility.
At West Bengal Chemical Industries Limited (WBCIL), this discipline is not something we built for inspection purposes. It is how we have operated for last 64 years.
Over six decades of manufacturing Active Pharmaceutical Ingredients (APIs) across pharmaceutical, nutraceutical, and food-grade categories means compliance is simply part of how things get done here.
This blog walks through what USFDA guidelines for pharmaceuticals actually require, why they matter for bulk drug manufacturers, and exactly how WBCIL meets — and in several areas exceeds — those standards in day-to-day production. Whether you are a procurement head evaluating API suppliers or a formulation scientist reviewing sourcing options, this should answer the questions that come up most.
Takeaways:
- Compliance Is a System, Not a Certificate
USFDA guidelines for pharmaceutical manufacturing — anchored in 21 CFR Parts 210 and 211 and ICH Q7 — require a living quality system, not a one-time audit pass. WBCIL’s WHO-GMP, ISO, and HACCP certifications are the visible outcome of a compliance culture built over 60+ years of pharmaceutical-grade API production. - Injectable API Purity Is Where Standards Are Hardest — and WBCIL Delivers
For injectable iron APIs like Ferric Carboxymaltose, WBCIL’s patented chlorine-free synthesis process achieves 96.88% purity while eliminating the genotoxic impurity risk that trips up other manufacturers. This is USFDA compliance in practice — not just claimed, but chemically proven. [3] - The Right API Supplier Accelerates Your Regulatory Journey
Sourcing from a USFDA-compliant API manufacturer like WBCIL reduces your submission timeline, simplifies incoming QC, and gives your supply chain the regulatory credibility that branded pharmaceutical buyers now routinely demand. Full documentation — CoAs, DMF support, ICH Q7-aligned records — is standard at WBCIL.

What Do USFDA Guidelines for Pharmaceutical Manufacturing Actually Cover?
Let us start with the basics, because there is a lot of confusion about what USFDA compliant actually means for an API manufacturer.
The core regulatory document is 21 CFR Parts 210 and 211 — the Current Good Manufacturing Practice (cGMP) regulations published by the US Food and Drug Administration. These are not optional standards or voluntary codes. Under Section 501(a)(2)(B) of the Federal Food, Drug and Cosmetic Act, all drugs — including APIs — must be manufactured in conformance with cGMP requirements. If they are not, they are considered adulterated, regardless of how good the end product looks on paper. [1][2]
For APIs there is a framework that is used everywhere. It is in ICH Q7. The Good Manufacturing Practice Guide for Active Pharmaceutical Ingredients. The International Council for Harmonisation or ICH published this guideline. The USFDA adopted it. It is like a guide for making APIs.
It covers things like
- Checking the materials you start with
- Controlling the process
- Making sure equipment works right
- Keeping data
- Tracking batches.
This guideline helps make sure APIs are made the way all, over the world.
What cGMP actually demands in practice?
So when we talk about a facility that makes Pharmaceutical Ingredients and follows the rules of good manufacturing practice it has to show proof that it does things the right way. The facility must be able to show documents that say:
- Every batch of Active Pharmaceutical Ingredients is made using steps that have been checked and written down
- The materials they use to make the Active Pharmaceutical Ingredients are tested and okayed before they are used
- The equipment they use is checked to make sure it works right. It is fixed and taken care of on a regular schedule
- If something does not go according to the process it is written down looked into and fixed
- The finished Active Pharmaceutical Ingredients are tested to make sure they meet the standards that have been agreed on before they are sent out
- All the records are complete, accurate. Can be tracked back, to where they came from. And nobody can change them after they have been written down.The Active Pharmaceutical Ingredients facility has to do all of these things to show that it is following the rules of manufacturing practice.
This is not a checklist that gets filed away after approval. It is an ongoing quality system that lives in the daily operation of the plant.
Regulatory Reference — USFDA (21 CFR Part 211)
“There shall be written procedures for production and process control designed to assure that the drug products have the identity, strength, quality, and purity they purport or are represented to possess.” — 21 CFR Part 211, Section 211.100 [1]
How WBCIL Maintains USFDA Standards in API Production?
WBCIL holds WHO-GMP and ISO certifications across its manufacturing facilities in Kolkata and Dahej, Gujarat. These are not entry-level credentials — WHO-GMP certification in particular is aligned with the USFDA’s own inspection expectations for overseas API manufacturers supplying the US market. But the certifications are the outcome, not the method. Here is how the compliance system actually works on the ground.
1. Quality Control at every stage of production
At WBCIL, quality control is not a department that reviews output at the end of a batch. It is embedded throughout production. Starting material testing, in-process controls, and finished product testing are all conducted against pharmacopoeia-grade specifications — USP (United States Pharmacopeia), BP (British Pharmacopoeia), and EP (European Pharmacopoeia) — depending on the intended market of the API.
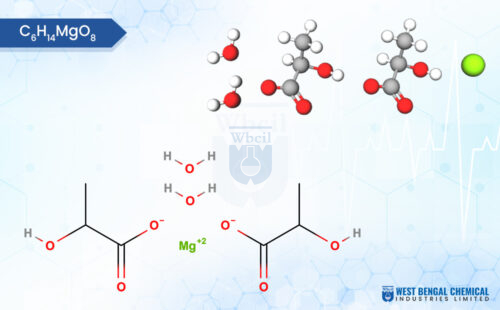
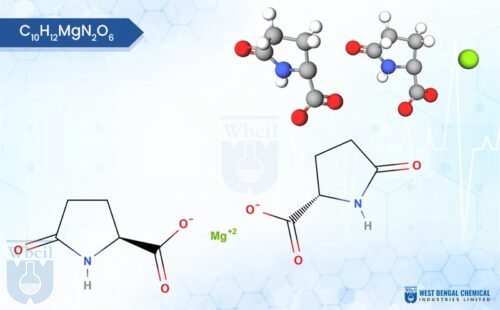
This means that a buyer sourcing WBCIL’s magnesium bisglycinate, ferric carboxymaltose, or zinc gluconate for a US-market product is receiving material that has been tested against the same identity, purity, and potency specifications that their finished-product QC team will use.
2. Data integrity and batch traceability
One of the most common reasons for USFDA warning letters to overseas API manufacturers is data integrity failures — records that were modified after the fact, results that were selectively reported, or audit trails that were disabled. This is treated with the same seriousness as a contamination failure, because it represents a fundamental breakdown in the quality system.
WBCIL maintains complete batch manufacturing records and laboratory notebooks in accordance with the ALCOA+ framework — Attributable, Legible, Contemporaneous, Original, Accurate — plus the extended principles of Complete, Consistent, Available and Enduring. Every batch is fully traceable from raw material certificate of analysis through to finished product release documentation.
3. Facility design and environmental controls
Both WBCIL facilities are designed with GMP-compliant architectural segregation — separate areas for weighing, synthesis, processing, and packaging, with appropriate HVAC, pressure differentials, and contamination controls for each product class. Utilities including water for pharmaceutical use, compressed air, and nitrogen are validated and tested on defined schedules.
WBCIL Certification Stack
WHO-GMP Certified | ISO 9001 & ISO 22000 | HACCP | FSSAI | cGMP Compliant Manufacturing at Kolkata and Dahej (Gujarat) | Exports to 35+ countries including regulated markets in the US, EU, and Middle East
Navigating USFDA Regulatory Requirements for Injectable APIs
Injectable APIs occupy a different compliance tier from oral-grade materials. The FDA’s expectations for sterile and parenteral-grade APIs are substantially more demanding — and rightly so, because anything administered intravenously bypasses every natural defence the body has against contamination.
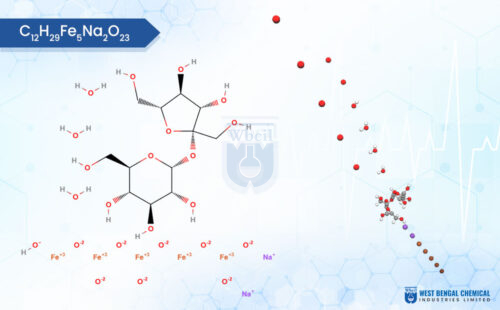
WBCIL is one of India’s few API manufacturers with deep specialisation in injectable iron compounds — a category that includes Ferric Carboxymaltose (FCM), Iron Isomaltoside, and Ferric Derisomaltose. These are complex carbohydrate-iron formulations used in clinical settings to treat iron deficiency anaemia, particularly in patients who cannot tolerate or have not responded to oral iron therapy.
The impurity challenge in injectable iron manufacturing
Traditional synthesis routes for iron carbohydrate complexes have historically used sodium hypochlorite as an oxidising agent. The problem is that hypochlorite can produce chlorinated organic by-products — genotoxic impurities that fall under ICH M7 guidelines and require strict control in pharmaceutical products intended for human use. For injectable formulations, even trace levels of these compounds represent a real safety concern. [3]
WBCIL addressed this directly through its patented process (Indian Patent 370845), which eliminates hypochlorite entirely from the synthesis route and replaces it with a citric acid-based stabilisation approach. The result is a Ferric Carboxymaltose API that achieves 96.88% purity with a 92% process yield — and without the chlorinated impurity profile that complicates regulatory submissions for injectable iron products.
WBCIL FCM Purity Data — Patent 370845
Purity: 96.88% | Process Yield: 92% | Iron Content: 34.55% |
Mean Molecular Weight: 82,000 Da | Less than 10% of complex below 10,000 Da — preventing toxicity and ensuring safe iron particle formation. No chlorinated organic by-products. [3]
Ensuring Trace Element Purity Under USFDA Guidelines
Trace elements — chromium, manganese, copper, zinc, selenium, cobalt — sit in a unique regulatory space. They are essential nutrients at physiological doses, but they carry potential toxicity at elevated concentrations, and their impurity profiles require careful monitoring during manufacturing.
For API manufacturers supplying trace elements in pharmaceutical-grade or nutraceutical formulations, this means running heavy metal testing (per USP <233> and <231>), microbiological testing, and residual solvent analysis as standard practice — not as exception testing triggered only by complaints.
WBCIL’s trace element portfolio and testing standards
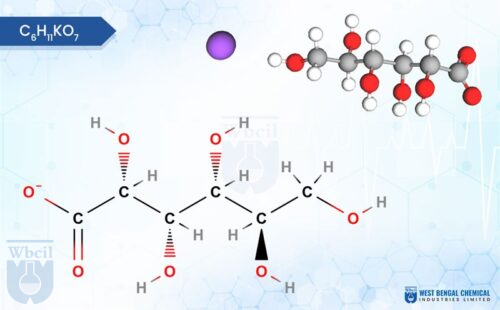
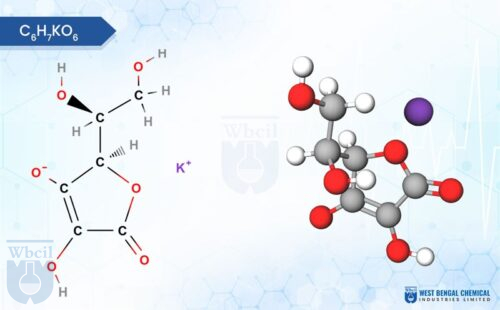
WBCIL produces a comprehensive range of mineral salts used as APIs and nutraceutical raw materials, including chromium picolinate, manganese gluconate, copper sulphate, and zinc bisglycinate, among others. All are manufactured to Pharmacopoeia-grade specifications and tested using validated analytical methods.
For products entering regulated markets — including the US, EU, and Gulf Cooperation Council — Certificates of Analysis (CoAs) are provided with full analytical data covering identity, assay, impurity profile, heavy metals, residual solvents (where applicable), microbial limits, and particle size where relevant. These documents are the primary tool buyers use to verify USFDA compliance at incoming QC.
WBCIL also maintains a product master file system, meaning that all quality documentation — specifications, manufacturing procedures, stability data, and analytical validation reports — is organised and accessible for regulatory submissions, including Drug Master File (DMF) support for US-market drug applications.
Benefits of Sourcing from a USFDA-Compliant API Manufacturer Like WBCIL
This might sound obvious, but it is worth spelling out. The commercial benefits of sourcing APIs from a USFDA-compliant manufacturer go beyond simply clearing customs.
Shorter regulatory submission timelines
When your API supplier provides a complete, well-organised regulatory package — CoA, DMF reference, ICH Q7-aligned documentation — your own drug product submission to the FDA moves faster. Gaps in supplier documentation are one of the leading reasons for information requests and delays in 505(b)(2) and ANDA filings.
Reduced incoming QC burden
A buyer who trusts the quality system behind their API supplier can operate with a reduced retesting protocol at incoming inspection. This is not about skipping tests — it is about having sufficient confidence in the supplier’s validated process to focus your own resources on finished product testing where it matters most.
Supply chain credibility for your end customers
Whether you are a contract manufacturer supplying a branded pharmaceutical company or a finished-dose brand selling into the US market, your upstream supply chain is increasingly subject to audit. Being able to document that your APIs come from a WHO-GMP and cGMP-compliant facility with a clean regulatory track record is a direct competitive advantage.
Lower risk of supply disruption
The Food and Drug Administration warning letters and import alerts and facility bans are not something that you hear about they are real. The Food and Drug Administration warning letters and import alerts and facility bans actually happen to companies that supply Active Pharmaceutical Ingredients.. When the Food and Drug Administration warning letters and import alerts and facility bans happen they cause big problems, for the companies that buy Active Pharmaceutical Ingredients from them. These problems can last for months.
If a supplier of Active Pharmaceutical Ingredients has done a job of following the Good Manufacturing Practice rules for a long time then it is safer to buy Active Pharmaceutical Ingredients from them. This is especially true for Active Pharmaceutical Ingredients that’re very important and hard to get from other companies.
To Sum It Up
Regulatory compliance in pharmaceutical manufacturing is one of those topics that can get buried under acronyms and framework references until it loses all meaning. But what it really comes down to is simple: can you prove — with documented evidence, on any given day — that your product is exactly what you say it is?
That question drives everything WBCIL does in its API manufacturing operations. From the written procedures governing every synthesis step, to the pharmacopoeia-grade testing that clears every batch for release, to the patented chemistry that eliminates impurity risks in injectable iron — compliance at WBCIL is not a posture. It is a practice.
For buyers sourcing bulk drugs and mineral APIs for regulated markets, the question is never just whether a supplier is certified. It is whether that supplier’s quality system actually delivers consistent, documented, audit-ready compliance — batch after batch, year after year.
After more than six decades and 35+ export markets, that is precisely what WBCIL is built to deliver.
Partner with WBCIL for USFDA-Aligned API Supply
West Bengal Chemical Industries Limited | www.wbcil.com |
WHO-GMP & ISO Certified | Kolkata & Dahej, India |
60+ Years of Pharmaceutical API Excellence |
Injectable Iron · Mineral Salts · Chelated APIs · Liposomal Ingredients Exports to 35+ Countries
- U.S. Food and Drug Administration. “Current Good Manufacturing Practice (cGMP) Regulations — 21 CFR Parts 210 and 211.” Updated June 2024.
- ICH Harmonised Guideline. “Q7: Good Manufacturing Practice Guide for Active Pharmaceutical Ingredients.” International Council for Harmonisation. Published 2000; adopted by USFDA as guidance for industry.
- U.S. FDA. “Questions and Answers on Current Good Manufacturing Practice Regulations — Production and Process Controls.” fda.gov
Eurofins USA. “Ensuring Drug Quality: An Overview of 21 CFR Parts 210 & 211.”
The USFDA’s pharmaceutical manufacturing guidelines are primarily set out in 21 CFR Parts 210 and 211 — the Current Good Manufacturing Practice (cGMP) regulations. For API manufacturers specifically, the globally harmonised ICH Q7 guideline provides the detailed framework for quality systems, process controls, batch documentation, and testing requirements. Non-compliance with these standards means an API can be classified as adulterated under the Federal Food, Drug and Cosmetic Act.
A GMP-certified mineral API production facility operates under documented, validated manufacturing processes with full batch traceability, qualified personnel, calibrated equipment, and approved testing methods. Every batch is released against pharmacopoeia-grade specifications. For mineral APIs — zinc, magnesium, iron, calcium, chromium, etc. — this includes identity testing, assay, heavy metal limits, microbiological testing, and in some cases particle size and dissolution profiling.
WBCIL operates WHO-GMP and ISO-certified facilities in Kolkata and Dahej with embedded quality systems — not just audit-ready documentation. This includes validated production procedures, data integrity controls aligned with the ALCOA+ framework, in-process and finished-product testing against USP/BP/EP standards, and full batch record retention. For injectable APIs like Ferric Carboxymaltose, WBCIL also holds patents for improved synthesis routes that eliminate genotoxic impurity risk.
Iron carbohydrate complexes like Ferric Carboxymaltose are parenteral-grade APIs that must meet extremely stringent purity requirements. Under USFDA and ICH M7 guidelines, genotoxic impurities must be controlled at very low levels. WBCIL’s patented chlorine-free synthesis process specifically eliminates the chlorinated organic by-products that form a key regulatory concern in traditional iron carbohydrate synthesis. The result is an FCM API with documented 96.88% purity and full ICH compliance.
Sourcing from a USFDA-compliant API manufacturer like WBCIL reduces regulatory submission timelines, simplifies incoming QC protocols, and provides supply chain documentation that stands up to pharmaceutical buyer audits. It also reduces the risk of supply disruption — FDA Warning Letters and Import Alerts can freeze API supply from non-compliant facilities for extended periods. For critical APIs, this supply security is as commercially important as the technical specification.
WBCIL maintains organised product documentation — specifications, manufacturing procedures, analytical validation reports, and stability data — that can support DMF filings for US market drug product applications. Buyers sourcing WBCIL APIs for regulated market formulations are encouraged to discuss documentation requirements with WBCIL’s technical team early in the procurement process.